The Weekly Term Sheet (2026-W10)

Visual Summary
Week in Numbers: $16.6B total disclosed value, $9.3B M&A, $295M+ royalty and non-dilutive capital, $838M equity financings, $8.2B debt issuances
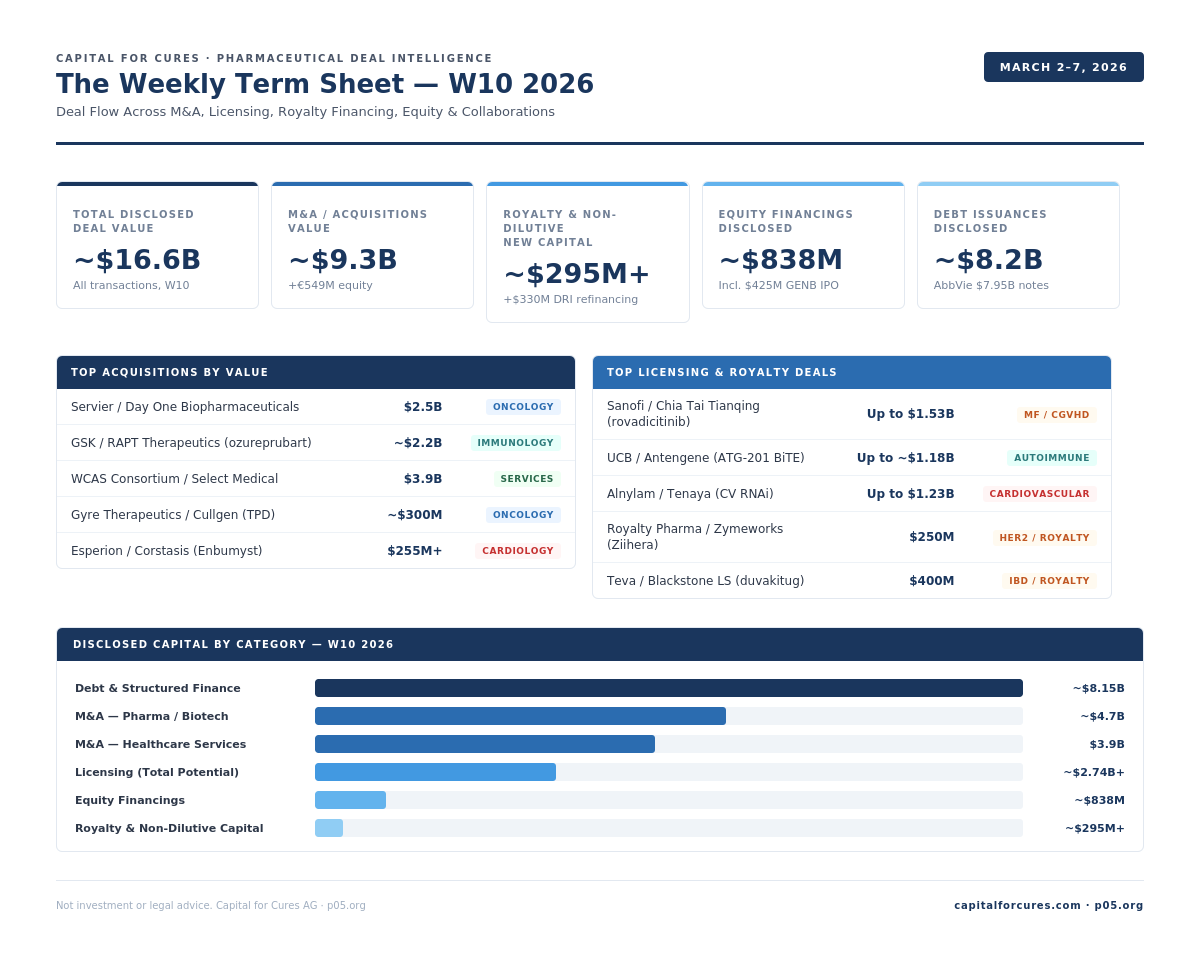
Royalty League Table: Seven royalty and non-dilutive financing transactions: Teva/Blackstone ($400M), Royalty Pharma/Zymeworks ($250M), Heidelberg/HCRx+Soleus ($45M), and DRI Healthcare ($330M+ refinancing)

M&A and Licensing League Tables: WCAS/Select Medical $3.9B through Esperion/Corstasis $255M+) and licensing ranking (Chia Tai Tianqing/Sanofi $1.53B through Pierre Fabre/Eton Pharma

FDA and EMA Regulatory Overview: 3 FDA approvals, 2 label expansions, 8 EMA positive opinions, 3 EMA negative/withdrawn, 2 FDA BTD/510k)

Week in Review
Week 10 produced two transactions with direct implications for the pharmaceutical royalty and financing ecosystem: AstraZeneca's royalty monetization with Compugen for rilvegostomig surfacing in earnings disclosures, and Teva's $400M strategic growth capital agreement with Blackstone Life Sciences for duvakitug — the week's most direct example of a dedicated life sciences royalty investor providing development capital in exchange for a future royalty interest. Together they illustrate two sides of the royalty deal spectrum: monetising receivables during Phase 3, and creating new royalty obligations to fund pipeline advancement.
EMS/NC Group's $600M acquisition of Sanofi's Brazilian generics unit Medley (signed March 6) was the week's most significant Latin American pharmaceutical transaction and Sanofi's full exit from Brazilian generics. Other notable developments included UniQure's ~40% stock decline following FDA's rejection of its BLA filing plan for AMT-130, Novo Nordisk's €432M Irish factory expansion and its Hims & Hers platform deal (which sent Hims shares up ~40%), Bayer's $7.25B Roundup settlement receiving preliminary court approval, and DRI Healthcare's $330M+ in combined refinancing alongside a new $800M–$1B five-year deployment target against a ~$3B pipeline.
Total disclosed deal value this week: ~$16.6B
Feature: Moderna / Arbutus Biopharma–Genevant Sciences — Up to $2.25B LNP Patent Settlement
Moderna / Arbutus Biopharma–Genevant Sciences | Patent Settlement + License | LNP Delivery Technology | Platform | March 3, 2026
| Parameter | Detail |
|---|---|
| Announced | March 3, 2026 |
| Structure | Consent to judgment of infringement + global perpetual royalty-free license |
| Non-contingent payment | $950M lump sum, due July 8, 2026 |
| Contingent payment | $1.3B contingent on outcome of Moderna's Federal Circuit appeal (Section 1498 government-contractor immunity defense) |
| Total maximum | Up to $2.25B |
| Ongoing royalties | None — royalty-free license |
| Patents covered | U.S. 8,492,359; 9,364,435 (survived Moderna's IPR challenge at PTAB); 11,141,378; 9,504,651 (four LNP molar ratio / method patents) |
| Products licensed | Spikevax, mNEXSPIKE, mRESVIA, mCOMBRIAX, and all future infectious disease vaccines using SM-102 LNP |
| Proceeds allocation | Arbutus ~20% (net of litigation costs, subject to tiered low single-digit royalty cap; evaluating return of capital to shareholders in Q3 2026); Genevant/Roivant ~80% |
| Scope | Global, non-exclusive, perpetual, irrevocable |
| Court | Arbutus Biopharma Corp. v. Moderna Inc., D. Del., No. 22-cv-252; consent judgment issued March 4, 2026 |
| Arbutus legal counsel | Morrison & Foerster (Adam Brausa, Eric Wiener, Daralyn Durie); Shaw Keller LLP (Delaware local) |
| Genevant legal counsel | Williams & Connolly (David Berl, lead); Shaw Keller LLP (Delaware local) |
| Moderna legal counsel | Kirkland & Ellis (James Hurst, Jeanna Wacker); Morris, Nichols, Arsht & Tunnell (Delaware local) |
| Roivant capital return | $1.0B share buyback authorized post-settlement |
Announced March 3, 2026, this settlement resolves all worldwide LNP patent enforcement actions between Moderna and Arbutus/Genevant — potentially the largest disclosed patent settlement in pharmaceutical industry history. The original 2022 complaint asserted six patents; the case narrowed to four by trial, all covering LNP molar ratios and formulation methods. Moderna consented to judgment of infringement on all four, receiving in return a global, non-exclusive, perpetual, royalty-free license covering its entire infectious disease vaccine portfolio using SM-102 lipid nanoparticle technology. The consent judgment was issued March 4, 2026 by Judge Joshua Wolson (E.D. Pa., sitting by designation in D. Del.).
The $950M non-contingent payment is due July 8, 2026. The additional $1.3B is contingent on the outcome of Moderna's Federal Circuit appeal of the Section 1498 government-contractor immunity defense — a legal theory under which Moderna argued that its COVID-19 vaccine production, carried out under U.S. government contracts, was shielded from patent infringement claims. Judge Wolson ruled February 4, 2026 that Section 1498's "for the Government" language requires actual government benefit, not merely public benefit through patients, preserving Moderna's right to appeal. If the Federal Circuit affirms, the $1.3B is due within 90 days; if Moderna ultimately prevails, Arbutus/Genevant must refund the full amount plus interest. Moderna has recorded no accrual for this contingency.
The proceeds split is asymmetric: Arbutus receives approximately 20% of net settlement proceeds (after litigation costs), subject to a cap of tiered low single-digit royalties on net sales, while Genevant/Roivant receives approximately 80%. Roivant authorized a $1.0B share buyback following the announcement.
The elimination of ongoing royalties is commercially significant: Moderna had faced the prospect of paying royalties on all future SM-102 LNP-based vaccine sales — a potentially multi-billion-dollar long-term liability — in exchange for the lump-sum resolution. Arbutus and Genevant exchanged that royalty stream for immediate capital certainty. The structure is a clean royalty monetization in reverse: the royalty obligor (Moderna) paid to extinguish rather than the royalty holder selling forward.
Feature: AstraZeneca / Compugen Royalty Monetization — rilvegostomig
AstraZeneca / Compugen | Royalty Financing | Lung, GI, and Endometrial Cancers | Phase 3
| Parameter | Detail |
|---|---|
| Upfront | $65M |
| Additional milestone (BLA acceptance) | $25M |
| Total maximum disclosed | $90M |
| Royalty rate retained by Compugen | Up to mid-single digits (majority retained) |
| Remaining milestone eligibility | Up to $195M |
| Deal announced | December 17, 2025 |
| Earnings confirmation | March 2, 2026 |
The AstraZeneca/Compugen deal originally closed in December 2025 but came into focus this week through Compugen's Q4 2025 earnings disclosure on March 2, confirming receipt of the $65M upfront payment. The structure is a canonical royalty monetization: Compugen sold a small portion of its future royalty interest in rilvegostomig — AstraZeneca's PD-1/TIGIT bispecific — in exchange for non-dilutive upfront capital, while retaining the majority of the royalty stream and all future milestone eligibility.
Rilvegostomig is an Fc-reduced PD-1/TIGIT bispecific antibody currently advancing across 10 ongoing Phase 3 trials in patients with lung, gastrointestinal, and endometrial cancers. The TIGIT component derives from Compugen's wholly owned COM902 antibody, licensed to AstraZeneca under a 2018 agreement. For Compugen, the transaction extended the expected cash runway to 2029 with no debt incurred, and the company turned profitable in 2025 — reporting $72.8M in full-year revenues versus $27.9M in 2024 — largely on the back of the AstraZeneca payment and earlier Gilead license fees.
The deal is structurally notable for the retained economics. Compugen gave up a minority slice of royalty flow to access immediate capital during Phase 3, rather than monetizing the entire royalty interest. As rilvegostomig approaches potential approval and commercialization, the residual royalty position could represent significant value — the company separately holds up to $758M in milestones from its Gilead partnership for GS-0321.
The term sheet value cited in the source data ($260M total) appears to reflect an estimate of broader royalty value rather than the $90M maximum disclosed in the official agreement. The $65M upfront and up to $195M in future milestones are the confirmed contractual figures.
Royalty Financing: Teva / Blackstone Life Sciences — duvakitug | $400M
Teva Pharmaceuticals / Blackstone Life Sciences | Royalty Financing / Strategic Growth Capital | Inflammatory Bowel Disease (Ulcerative Colitis, Crohn's Disease) | Phase 3
| Parameter | Detail |
|---|---|
| Announced | March 3, 2026 |
| Financing amount | $400M, disbursed over four years |
| Funder | Blackstone Life Sciences (BXLS) |
| Asset | Duvakitug (TEV-'574 / SAR447189), anti-TL1A human monoclonal antibody |
| Indications | Ulcerative colitis (SUNSCAPE-1), Crohn's disease (STARSCAPE-1) |
| Phase 3 enrollment | >3,000 patients combined; follow-up up to 40 weeks |
| Phase 3 completion | SUNSCAPE-1 (UC): expected 2028; STARSCAPE-1 (CD): expected 2029 |
| BXLS consideration | FDA-approval milestone (undisclosed size) + commercial milestones + low single-digit royalties on worldwide net sales |
| Royalty rate | Low single-digit (global, all territories) |
| Co-development partner | Sanofi (separate October 2023 agreement; equal cost/P&L share in major markets; Sanofi leads Phase 3 and North America/Asia commercialization; Teva leads Europe/Israel) |
| Sanofi upfront to Teva (2023) | €469M (~$500M) |
| Sanofi milestones to Teva (2023) | Up to €940M (~$1B) in development and launch milestones |
| Phase 2b LTE data | Reported February 17, 2026: 58% UC patients, 55% CD patients in clinical remission at 44 weeks maintenance |
| Key BXLS spokespeople | Paris Panayiotopoulos, Senior Managing Director, BXLS; Dr. Nicholas Galakatos, Global Head, BXLS |
| Key Teva spokesperson | Evan Lippman, EVP Business Development, Teva |
| BXLS legal counsel | Ropes & Gray LLP |
| Teva legal counsel | Not disclosed |
| Financial advisors | Not disclosed |
Announced March 3, 2026, Teva's agreement with Blackstone Life Sciences provides $400M in four-year staged funding to cover ongoing and future duvakitug development costs, in exchange for a contingent FDA-approval milestone payment, additional commercial milestones, and low single-digit royalties on worldwide net sales. Ropes & Gray LLP acted as legal counsel to Blackstone Life Sciences — consistent with the firm's standing role advising BXLS on its life sciences financing transactions, having also represented BXLS on its Moderna and Alnylam growth capital agreements. Teva's legal counsel and any financial advisors were not disclosed in the public announcement.
The asset: duvakitug and TL1A biology
Duvakitug is a fully human monoclonal antibody targeting TL1A (TNF-like cytokine 1A, also called TNFSF15), a member of the TNF superfamily expressed primarily by macrophages, dendritic cells, and endothelial cells in inflamed tissue. TL1A drives both inflammatory and fibrotic pathways in the intestinal wall through its receptor DR3 (Death Receptor 3), which is expressed on T cells, innate lymphoid cells, and stromal cells. The dual mechanism distinguishes TL1A inhibition from anti-TNF, IL-12/23, and gut-selective integrin therapies, which primarily suppress inflammatory cascades but have limited impact on the fibrotic process that drives stricturing disease, obstruction, and surgical intervention.
In Phase 2b, duvakitug at 900mg demonstrated clinical remission in 58% of UC patients and 55% of CD patients at the end of a 44-week maintenance period — durability data reported February 17, 2026, two weeks before the Blackstone financing. Phase 3 comprises two studies: SUNSCAPE-1 (ulcerative colitis, expected data 2028) and STARSCAPE-1 (Crohn's disease, expected data 2029), together enrolling over 3,000 patients.
The competitive TL1A landscape
Duvakitug is not alone in Phase 3. Merck & Co. acquired tulisokibart via its $10.8B purchase of Prometheus Biosciences in 2023 and expects Phase 3 data later in 2026. Roche's afimkibart, acquired as part of a $7B deal for Telavant, is also in pivotal testing.
AbbVie is developing an additional TL1A program. The class is therefore crowded at the most critical juncture, and payer positioning — step therapy placement relative to anti-TNF, IL-23, JAK inhibitors, and gut-selective agents — will depend heavily on head-to-head data or differentiated trial design that Phase 3 may or may not deliver.
The prior Sanofi collaboration and the stacked royalty architecture
In October 2023, Sanofi paid €469M (~$500M) upfront to co-develop and co-commercialize duvakitug globally, with up to €940M in additional development and launch milestones to Teva. The agreement splits global development costs equally, shares major-market net P&L on a 50/50 basis, and designates Sanofi as Phase 3 lead and North American/Asian commercial lead, with Teva leading European and Israeli commercialization. Non-major markets operate under a separate royalty arrangement between Teva and Sanofi.
The BXLS low single-digit global royalty now sits on top of this co-commercialization structure. Blackstone's royalty runs on worldwide net sales regardless of territory, meaning it applies across both the Sanofi-led and Teva-led commercial geographies simultaneously. The combined effect is a layered royalty burden: in North American and Asian markets, Sanofi's 50/50 P&L split and Teva's royalty obligation to BXLS both reduce Teva's net economic participation; in European and Israeli markets where Teva leads commercialization, the BXLS global royalty still applies as a first claim on revenue.
Analysts have flagged that this stacked structure compresses net pricing power in formulary negotiations, particularly against IL-23 inhibitors that reach the market with lighter royalty stacks. The undisclosed size of the FDA-approval milestone owed to BXLS adds a further contingent capital commitment at the point of regulatory success.
BXLS's strategic pattern
Blackstone Life Sciences has signed structurally similar development capital agreements with Moderna, Alnylam Pharmaceuticals, and Autolus Therapeutics — a pattern of providing staged research and development funding to Phase 3-stage assets in exchange for royalties and milestone-linked returns rather than equity ownership. The model functions as a hybrid between venture lending and royalty acquisition, providing pharmaceutical companies with non-dilutive capital while creating royalty-like economic exposure for the investor without requiring asset ownership. The Teva/duvakitug transaction extends this model to a co-commercialized asset, creating a three-party royalty structure: BXLS (global royalty), Sanofi (major-market P&L split), and Teva (residual after both).
The duvakitug deal is Teva's second consecutive non-dilutive pipeline financing in early 2026. In January 2026, Teva secured up to $500M from Royalty Pharma to fund the clinical development of TEV-'408, an anti-IL-15 monoclonal antibody in Phase 2 for celiac disease and Phase 1b for vitiligo. The back-to-back transactions — Royalty Pharma in January, BXLS in March — reflect Teva's stated "Pivot to Growth" capital strategy: funding Phase 2 and Phase 3 pipeline advancement through royalty-based and growth capital instruments rather than equity dilution or balance sheet drawdown. Both deals create royalty obligations that will sit above Teva's net sales economics if either asset reaches market.
Acquisitions
| # | Acquirer | Target / Asset | Value | Type | Stage | Date |
|---|---|---|---|---|---|---|
| 1 | Servier | Day One Biopharmaceuticals (Ojemda + pipeline) | $2.5B | Cash tender offer | Marketed / Ph 3 | March 6 |
| 2 | Rallybio | Candid Therapeutics (T-cell engager platform) | Undisclosed + CVR | Reverse merger + financing | Ph 1 / Ph 2 | March 3 |
| 3 | Gyre Therapeutics | Cullgen (protein degradation platform) | ~$300M all-stock | All-stock | Clinical | March 2 |
| 4 | Esperion | Corstasis (Enbumyst) | $75M upfront + up to $180M milestones + royalties | Cash + royalty obligation | Marketed | March 3 |
| 5 | GSK | RAPT Therapeutics (ozureprubart) | ~$2.2B (~$1.9B net cash) | Cash tender offer completion | Ph IIb | March 3 |
| 6 | RadNet / DeepHealth | Gleamer SAS (radiology AI) | Up to €230M | Cash + milestone | Marketed | March 2 |
| 7 | WCAS Consortium | Select Medical Holdings | $3.9B | Take-private | Commercial | March 2 |
| 8 | Grünenthal | Kyowa Kirin International EMEA JV stake | Undisclosed | Stake buyout | Commercial | March 2 |
| 9 | Intuitive Surgical | ab medica / Abex / Excelencia Robótica | ~€319M | Completion | Commercial | March 1–2 |
| 10 | Sectra | Oxipit (chest X-ray AI) | Undisclosed | Acquisition | Marketed | March 2026 |
| 11 | BioSyent | Oral Science | $25.5M + up to $6M contingent royalties | Cash + equity + royalty | Commercial | March 2 |
| 12 | VERAXA Biotech | Voyager Acquisition Corp. | Undisclosed | SPAC shareholder approval | Clinical | W10 |
Section total (disclosed values): ~$9.3B cash/equity + €549M + undisclosed elements. Servier/Day One ($2.5B) and GSK/RAPT (~$2.2B) account for ~$4.7B of disclosed pharma deal value. Select Medical ($3.9B) is healthcare services.
Royalty notes: Servier/Day One carries a layered royalty structure (mid-teens Ipsen receivable ex-U.S. inbound; undisclosed Takeda liability on U.S. sales outbound). BioSyent/Oral Science carries a seller-retained contingent royalty capped at $6M through 2033 on one undisclosed product (rate not disclosed). GSK/RAPT: no royalty obligation disclosed; Esperion/Corstasis: Otsuka Japan royalty (15–30% tiered) being monetized via Athyrium/HCRx to finance acquisition; Corstasis sellers receive low double-digit royalties on worldwide Enbumyst net sales going forward.
Acquisition: Servier / Day One Biopharmaceuticals — Ojemda + Oncology Pipeline | $2.5B
Servier / Day One Biopharmaceuticals | Acquisition (Cash Tender Offer) | Pediatric Low-Grade Glioma, Rare Oncology (Adult and Pediatric) | Marketed / Phase 3 | March 6, 2026
| Parameter | Detail |
|---|---|
| Announced | March 6, 2026 |
| Deal value | $2.5B (total equity value) |
| Price per share | $21.50 per share, all-cash |
| Premium to last close (March 5, 2026) | ~68% |
| Premium to 1-month VWAP (March 5, 2026) | ~86% |
| Structure | Cash tender offer; second-step merger for any untendered shares at same consideration |
| Closing conditions | Majority tender of DAWN shares; U.S. antitrust (HSR) clearance |
| Expected close | Q2 2026 |
| Financing | Servier existing cash and investments (no financing condition) |
| Servier advisors | Goldman Sachs Bank Europe SE (financial); Baker McKenzie (legal) |
| Day One advisors | Centerview Partners LLC (financial); Fenwick & West LLP (legal) |
| Day One ticker | NASDAQ: DAWN |
Announced March 6, 2026, Servier's acquisition of Day One Biopharmaceuticals is the largest biopharma deal announced this week, and the most significant oncology acquisition by a European private pharmaceutical company in recent years. Servier is a French foundation-governed group with €6.9 billion in revenues in the 2024/25 financial year, more than 20,000 employees, and medicines distributed across 130 countries. Despite its scale, it is privately held and foundation-governed — not publicly traded — making its deployment of $2.5 billion in existing balance sheet cash a significant strategic commitment to its oncology build.
The deal centerpiece is Ojemda (tovorafenib), a RAF kinase inhibitor that received FDA approval in April 2024 as the first oral, once-weekly targeted therapy for relapsed or refractory pediatric low-grade glioma (pLGG). pLGG is the most common form of brain cancer in children and adolescents; BRAF alterations — either the BRAF V600E mutation or BRAF fusions — drive approximately 50% of cases and have historically lacked approved oral targeted options. Ojemda's approval was based on the FIREFLY-1 Phase 2 study, which demonstrated a 67% overall response rate by independent review in BRAF-altered pLGG. The FDA granted Priority Review and Rare Pediatric Disease designation; a Pediatric Priority Review Voucher was awarded at approval.
Beyond Ojemda, the acquisition captures the pipeline assets Day One assembled through its January 2026 acquisition of Mersana Therapeutics: most notably XMT-2056, an antibody-drug conjugate targeting HER2 in advanced solid tumors, which entered Phase 1 in 2024 and represents a differentiated ADC payload mechanism. Day One's full pipeline spans early stage through Phase 3, with programs covering both pediatric and adult rare cancers — a strategic fit with Servier's stated 2030 objective of building leadership in rare oncology targeted therapies.
The premium — 68% to last close, 86% to the 30-day VWAP — is consistent with control premiums in competitive rare disease oncology acquisitions and reflects both the near-term commercial ramp of Ojemda and the option value in the broader pipeline. DAWN shares rose approximately 65% on announcement day, confirming a clean arb setup for the cash tender.
Goldman Sachs Bank Europe SE acted as exclusive financial advisor to Servier, with Baker McKenzie as legal counsel. Centerview Partners served as Day One's exclusive financial advisor, with Fenwick & West LLP as legal counsel.
Royalty Architecture
The Servier/Day One transaction carries a layered royalty structure that is material to understanding the deal's economics. Servier is simultaneously acquiring a royalty receivable and assuming a royalty liability — two flows running in opposite directions through the same asset.
Downstream — Ipsen royalty receivable (ex-U.S.): In July 2024, Day One licensed all ex-U.S. commercialization rights for tovorafenib to Ipsen for $111M upfront (including a $40M equity investment), up to $350M in milestone payments, and tiered double-digit royalties starting at mid-teens percentage on net sales. The royalty obligations run on a country-by-country basis for no less than ten years from first commercial sale in each territory. Servier acquires this receivable in full — Ipsen will pay mid-teens royalties on every dollar of ex-U.S.
Ojemda revenue directly to Servier going forward. The timing is favorable: Ojemda received a positive CHMP opinion from the EMA in February 2026, meaning the European royalty stream is about to become commercially live, potentially in the months immediately following deal close. No third-party royalty fund (Royalty Pharma, HCRx, OMERS, DRI Healthcare, or similar) holds a purchased interest in this stream — Day One never monetized the Ipsen receivable, so it transfers to Servier intact.
Upstream — Takeda royalty liability (U.S.): Tovorafenib was originally discovered in a Sunesis Pharmaceuticals / Biogen Idec collaboration that later expanded to include Millennium Pharmaceuticals, a Takeda affiliate. Day One acquired Takeda's rights in the compound in 2020 under a license from Takeda Oncology. That license almost certainly carries upstream royalty obligations on U.S.
Ojemda net sales payable back to Takeda — and potentially residual obligations to Biogen's successors or former Sunesis holders. The specific rate has never been publicly disclosed. Servier assumes these upstream obligations with the acquisition.
Industry analysts flagged this liability at the time of Ojemda's FDA approval: the exact royalty owed to Biogen, Takeda, Viracta, and/or former Sunesis holders remains an open question, and the molecule's lengthy pre-commercialization development history means multiple parties may hold a claim on the revenue stack. The net economic spread — mid-teens ex-U.S. inbound from Ipsen, offset by an undisclosed upstream rate outbound to Takeda on U.S. sales — is a key variable in Servier's actual Ojemda return on invested capital that the $2.5B headline does not surface.
Acquisition / Reverse Merger: Rallybio / Candid Therapeutics — T-Cell Engager Autoimmune Platform
Rallybio Corporation / Candid Therapeutics | Acquisition / Reverse Merger + Concurrent Financing | Autoimmune Diseases (Myasthenia Gravis, ILD, SLE, RA, and others) | Phase 1 / Phase 2
| Parameter | Detail |
|---|---|
| Announced | March 2, 2026 |
| Structure | All-stock reverse merger (Rallybio acquires Candid) |
| Concurrent financing | >$505M (oversubscribed and upsized) |
| Pro-forma cash at close | ~$700M |
| Runway | Through 2030 |
| Expected close | Mid-2026 |
| Post-merger name | Candid Therapeutics, Inc. |
| Post-merger ticker | NASDAQ: CDRX |
| Ownership split | Candid/new investors ~96.35%; legacy Rallybio ~3.65% |
| Rallybio CVRs | Tied to proceeds from legacy asset monetization (REV102 + others) |
| Key investors | RA Capital Management, Venrock Healthcare Capital Partners, Cormorant Asset Management, Foresite Capital, Janus Henderson Investors |
| Candid advisors | Wedbush Securities (financial); Cooley LLP (legal) |
| Rallybio advisors | Evercore (lead financial); Citizens Capital Markets & Advisory (co-financial); Ropes & Gray LLP (legal) |
| Placement agents ($505M financing) | Jefferies, BofA Securities, TD Cowen, Cantor Fitzgerald |
| Placement agent legal counsel | Latham & Watkins LLP |
Announced March 2, 2026, the Rallybio/Candid transaction is a classic reverse merger used to take a well-funded private clinical-stage company public: Rallybio (NASDAQ: RLYB), a New Haven-based rare disease biotech with a diminished pipeline following the Phase 2 failure of its lead FNAIT program in 2024, serves as the public shell, while Candid — the clinical and commercial substance — survives as the operating entity under its own name. The exchange ratio is determined by relative valuations at closing. The mechanics leave Rallybio stockholders with 3.65% of the combined company plus CVRs linked to monetization of legacy Rallybio assets; the remaining 96.35% accrues to Candid shareholders and the new financing round participants.
The concurrent $505M private financing was oversubscribed and upsized, with a syndicate of prominent healthcare institutional investors and mutual funds. The combined pro-forma cash position of approximately $700M is intended to fund operations through 2030, covering multiple Phase 2 readouts and pipeline advancement across Candid's T-cell engager (TCE) portfolio.
What Candid brings: T-cell engagers for autoimmune disease
Candid Therapeutics is a San Diego-based company that has assembled what it describes as the most advanced and diversified TCE pipeline in autoimmune disease. T-cell engagers are bispecific antibodies engineered to physically bridge a T cell (typically via a CD3-binding arm) to a pathogenic target cell, redirecting cytotoxic T-cell activity against disease-driving B cells or plasma cells — an approach that has proven transformative in hematologic oncology and is now being applied systematically to autoimmune disease. Candid's assets were acquired via two deals with Chinese biotechnology firms in 2024, giving the company a China-originated but globally rights-held portfolio.
The lead asset, cizutamig, is a BCMA-directed TCE. BCMA (B-cell maturation antigen) is expressed on long-lived plasma cells, which are the primary source of pathogenic autoantibodies in diseases such as myasthenia gravis, systemic lupus erythematosus, and IgG4-related disease. By depleting BCMA-expressing plasma cells, cizutamig aims to eliminate the autoantibody-producing cells responsible for disease maintenance.
With 87 total patients dosed including 47 autoimmune patients across more than 10 indications, cizutamig has demonstrated favorable tolerability with low rates of mild cytokine release syndrome. Phase 2 studies in myasthenia gravis and interstitial lung disease are planned for 2026.
CND261 is a CD19-directed TCE targeting B cells upstream of plasma cell differentiation, with initial clinical data expected in the first half of 2026. CND319 is a dual CD19/CD20 TCE designed to reduce antigen escape through dual B-cell surface targeting, with Phase 1 planned for mid-2026. CND460 is a preclinical dual BCMA/CD19 TCE expected to enter the clinic in the first half of 2027.
The autoimmune TCE space is rapidly crowding — Cullinan Therapeutics' CLN-978 (CD19 TCE) is approaching Phase 1 data in rheumatoid arthritis and SLE in 2026, and multiple other companies are pursuing overlapping targets. Candid's differentiation lies in the breadth of the pipeline, the clinical head start of cizutamig with data across a wide indication set, and the $700M capitalization enabling simultaneous multi-indication development.
The deal is included here for completeness as a major structural transaction in the autoimmune biologics space. It does not carry direct royalty financing implications at announcement, though the CVR structure for Rallybio legacy asset monetization (including REV102 rights) adds a royalty-adjacent element for legacy shareholders.
Acquisition: Gyre Therapeutics / Cullgen — Targeted Protein Degradation Platform | ~$300M All-Stock
Gyre Therapeutics / Cullgen | Acquisition | Fibrosis, Oncology, Pain | Clinical-Stage
| Parameter | Detail |
|---|---|
| Deal value | ~$300M (all-stock) |
| Structure | Tax-free reorganization; Cullgen becomes wholly owned subsidiary |
| Consideration | Gyre common stock + new Series B Convertible Preferred Stock (5:1 conversion ratio to common, post-stockholder approval; individual ownership capped below 19.99%) |
| Seller | Cullgen Inc. (privately held, San Diego) |
| Acquirer | Gyre Therapeutics, Inc. (Nasdaq: GYRE) |
| Leadership change | Cullgen CEO Dr. Ying Luo becomes President and CEO of combined company; Ping Zhang remains Executive Chairman; two Gyre directors resign at close |
| Regulatory conditions | HSR review; Cullgen stockholder approval; NYSE (GYRE) stockholder approval for share issuance |
| Expected close | Early Q2 2026 |
| Financial advisor | None disclosed |
| Announced | March 2, 2026 |
Gyre Therapeutics, a commercial-stage fibrosis-focused biopharma with $105.8M in 2024 net sales from ETUARY (pirfenidone) in China and a Hydronidone NDA approaching submission for CHB-associated liver fibrosis, agreed on March 2 to acquire Cullgen in a $300M all-stock deal. The transaction ends a prior reverse-merger agreement between Cullgen and Pulmatrix (terminated prior to signing) and delivers Cullgen's targeted protein degradation (TPD) and degrader-antibody conjugate (DAC) platform into Gyre's pipeline.
Cullgen's lead clinical asset is CG001419, an oral pan-TRK protein degrader targeting acute pain (post-bunionectomy Phase 2 planned for H1 2026) and solid tumors (Phase 1 ongoing). A second clinical-stage asset is a GSPT1 degrader for blood cancers. The DAC platform represents an emerging modality combining targeted degradation with antibody-directed delivery, extending the therapeutic scope well beyond Gyre's existing fibrosis franchise.
The combined entity will operate with U.S.- and China-based capabilities spanning discovery, manufacturing, and commercialization across inflammatory diseases, oncology, and pain. The all-stock structure preserves Gyre's cash for integration and pipeline investment. The Series B preferred share mechanism — designed to stay below 19.99% common stock issuance before stockholder conversion approval — is a standard structural solution for deals where dilution limits require bridging.
Acquisition: Esperion Therapeutics / Corstasis Therapeutics — Enbumyst | $255M + Royalties | CHF
Esperion Therapeutics / Corstasis Therapeutics | Acquisition | Edema / Congestive Heart Failure, Hepatic and Renal Disease | Marketed
| Parameter | Detail |
|---|---|
| Announced | March 3, 2026 |
| Upfront | $75M cash |
| Milestones | Up to $180M (regulatory and commercial) |
| Royalties to sellers | Low double-digit on worldwide Enbumyst sales and follow-on products |
| Total potential value | Up to $255M + ongoing royalties |
| Expected close | Q2 2026 |
| Esperion advisor | Jefferies LLC (financial); Gibson, Dunn & Crutcher LLP (legal) |
| Corstasis advisor | PJT Partners (financial); Arnold & Porter Kaye Scholer LLP (legal) |
| Financing | Esperion existing credit facilities + royalty monetization of Japanese bempedoic acid royalties via Athyrium Capital Management and HealthCare Royalty (KKR) |
| Japanese royalty payer | Otsuka Pharmaceutical Co., Ltd. (Japan-exclusive commercialization rights holder for NEXLETOL/NEXLIZET) |
| Japan royalty rate (underlying) | 15–30% tiered on net sales in Japan |
Announced March 3, 2026, Esperion Therapeutics (NASDAQ: ESPR) agreed to acquire all outstanding stock of Corstasis Therapeutics, a privately held commercial-stage company based in Henderson, Nevada, adding Enbumyst (bumetanide nasal spray) to its cardiovascular franchise. Enbumyst received FDA approval in September 2025 as the first and only nasal spray loop diuretic approved for edema associated with congestive heart failure, hepatic disease, and renal disease in adults. The stock fell 13.1% on announcement day, reflecting market skepticism about the cost of monetizing a newly commercial royalty stream to fund the deal.
Enbumyst: the clinical rationale
Loop diuretics — of which bumetanide is one — are the cornerstone of fluid management in heart failure, but existing delivery routes each have a meaningful limitation. Oral bumetanide and furosemide suffer from erratic gastrointestinal absorption in decompensated patients, precisely because gut wall edema impairs uptake when it is most needed. Intravenous administration is reliably effective but requires hospital or infusion center access, driving a significant proportion of CHF hospitalizations — patients who are fluid-overloaded at home but cannot achieve therapeutic diuresis orally cycle into the hospital for IV treatment and are discharged once stable.
Intranasal delivery bypasses the gut entirely, achieving faster and more consistent absorption than oral while remaining self-administered at home. Corstasis is also advancing a subcutaneous multidose pen injector pipeline as a further option for patients who cannot reliably self-administer nasal spray. Esperion cites a U.S. addressable market exceeding $4.6 billion, anchored by approximately 6.7 million American adults living with CHF.
The Otsuka Japan royalty stream — background
Otsuka is not the acquirer; it is the entity whose royalty payments to Esperion are being monetized to finance the deal. In April 2020, Esperion granted Otsuka Pharmaceutical Co., Ltd. exclusive rights to develop and commercialize NEXLETOL (bempedoic acid) and NEXLIZET (bempedoic acid/ezetimibe) in Japan, receiving a $60M upfront payment, up to $450M in development and sales milestones, and tiered royalties of 15–30% on all net sales in Japan. Otsuka was responsible for all Japan-specific development costs, regulatory submissions, and commercialization activities.
NEXLETOL received Japanese Ministry of Health, Labour and Welfare approval in September 2025 — just months before the Corstasis deal — with National Health Insurance pricing following shortly after. Otsuka deployed a dedicated field force of 600–800 sales representatives to support the launch. The Japan royalty stream was therefore brand new at the time Esperion chose to monetize it: commercially live for only a matter of months, with the full revenue ramp still ahead.
Athyrium Capital Management and HealthCare Royalty — who they are
Athyrium Capital Management is a New York-based specialized healthcare asset manager founded in 2008, with over $4.6 billion in committed capital. Athyrium invests across healthcare verticals — biopharma, medical devices, healthcare services, health IT — and across the capital structure, including royalties, structured debt, and equity. It has been an active participant in royalty monetization transactions for mid-cap commercial-stage biotechs, providing lump-sum liquidity against future royalty receivables in exchange for the right to collect those cash flows up to a negotiated return threshold, after which flows revert to the originator.
Athyrium's participation here signals the transaction was sized and structured as a conventional royalty purchase agreement layered on top of Esperion's existing credit facilities rather than a pure debt instrument.
HealthCare Royalty (HCRx) is a Greenwich, Connecticut-based royalty acquisition firm founded in 2006, majority owned by KKR & Co. (NYSE: KKR). HCRx has committed over $7 billion across more than 100 transactions in commercial-stage and near-commercial-stage biopharmaceutical royalties. It is one of the three dominant dedicated royalty acquirers in the life sciences space alongside Royalty Pharma and OMERS Life Sciences (which separately holds Esperion's European royalty stream monetized in July 2024 in a $304.7M transaction).
The co-participation of Athyrium and HCRx in the Japan monetization suggests the deal was either syndicated across both vehicles or structured as a co-investment — consistent with how larger royalty monetization facilities are often assembled when the underlying asset is a single-country royalty stream requiring diversification of funder exposure.
The circular royalty economy — one transaction, four layers
This deal is structurally notable for compressing the full pharmaceutical royalty lifecycle into a single announcement:
- Esperion holds a royalty receivable from Otsuka on Japan bempedoic acid net sales (15–30% tiered, newly commercial as of late 2025)
- Esperion monetizes a portion of that receivable to Athyrium/HCRx today, receiving a present-value lump sum in exchange for future Japan royalty cash flows up to an agreed return threshold
- Esperion deploys that lump sum as the $75M acquisition payment for Corstasis
- Corstasis shareholders receive in return low double-digit royalties on worldwide Enbumyst net sales going forward
The acquirer simultaneously holds a royalty receivable (Japan), converts it to present value via a royalty purchase, deploys the proceeds as acquisition currency, and creates a new royalty obligation to the sellers — all in one transaction. The Japan royalty stream is the collateral, the repayment mechanism, and the enabling instrument. The market's 13% negative reaction on announcement day likely reflects the cost of selling a newly-launched, high-potential royalty stream at early-commercial multiples to fund an acquisition in an adjacent but unproven indication, combined with the new ongoing royalty obligation to Corstasis shareholders reducing future Enbumyst free cash flow.
Acquisition Completion: GSK / RAPT Therapeutics — ozureprubart | ~$2.2B | Atopic Dermatitis / Food Allergy
GSK / RAPT Therapeutics | Acquisition Completion | Atopic Dermatitis; Food Allergy | Phase IIb | March 3, 2026
| Parameter | Detail |
|---|---|
| Transaction type | Tender offer completion — previously announced January 20, 2026 |
| Acquirer | GSK plc (LSE/NYSE: GSK) |
| Target | RAPT Therapeutics, Inc. (Nasdaq: RAPT) — delisted March 3, 2026 |
| Price | $58.00 per share (cash) |
| Equity value | ~$2.2B |
| Net of cash | ~$1.9B |
| Shares tendered | ~93% as of offer expiration |
| Delisting | Nasdaq; RAPT common stock ceased trading March 3, 2026 |
| Key asset | Ozureprubart (RPT193), long-acting anti-IgE monoclonal antibody |
| Mechanism | Anti-IgE; similar class to omalizumab (Xolair) but engineered for extended half-life and subcutaneous dosing at infrequent intervals |
| Lead indication | Food allergy prophylaxis (Phase IIb; data expected 2027) |
| Additional indication | Atopic dermatitis (Phase IIb) |
| Differentiation vs. omalizumab | Longer dosing interval (potential quarterly dosing vs monthly); designed for higher-affinity IgE suppression at lower doses |
| Strategic rationale | Bolt-on to GSK's immunology/allergy franchise; anti-IgE represents a validated mechanism; food allergy is an unmet need with no approved prophylactic oral or injectable agent |
| Original announcement | January 20, 2026 |
| Close | March 3, 2026 |
GSK closed its acquisition of RAPT Therapeutics on March 3, 2026, following a tender offer in which approximately 93% of shares were tendered at $58.00 per share. The transaction was originally announced January 20, 2026 and closed in approximately six weeks — a fast timeline consistent with the absence of major antitrust concerns and the straightforward structure of a cash tender offer for a clinical-stage company with no marketed products.
The strategic rationale centers on ozureprubart, a long-acting anti-IgE monoclonal antibody in Phase IIb for food allergy prophylaxis and atopic dermatitis. Anti-IgE is a validated mechanism — omalizumab (Xolair, Novartis/Genentech) has been approved since 2003 for allergic asthma and is now approved for IgE-mediated food allergies (2024) — but ozureprubart is engineered for a substantially extended dosing interval, potentially enabling quarterly subcutaneous administration versus the monthly schedule of omalizumab. If Phase IIb data in food allergy are positive (expected 2027), GSK would have a best-in-class convenience profile in a market where the approved Xolair label was only expanded in 2024. Food allergy affects approximately 32 million Americans, and there is no disease-modifying oral or injectable prophylactic agent approved beyond the omalizumab indication.
The $2.2B price tag for a Phase IIb-stage asset reflects both the validated mechanism and the scarcity value: the anti-IgE space is small, RAPT was the most advanced independent player, and GSK paid for optionality across two indications (food allergy and atopic dermatitis) with a single differentiated molecule.
Acquisition: RadNet / Gleamer SAS — Radiology AI, €230M | Digital Health / AI Imaging | Marketed
RadNet / Gleamer SAS | Acquisition | Radiology AI / Chest X-Ray, Thoracic CT | Marketed | March 2, 2026
| Parameter | Detail |
|---|---|
| Announced | March 2, 2026 |
| Deal value | Up to €230M, all-cash (including post-closing milestone) |
| Structure | Acquisition of 100% of Gleamer SAS; integration into RadNet's DeepHealth subsidiary |
| Target HQ | Paris, France |
| Acquirer | RadNet, Inc. (NASDAQ: RDNT), Woodland Hills, California |
| Customer base | Gleamer serves radiologists and radiology groups globally |
RadNet agreed to acquire Gleamer SAS, a Paris-based radiology AI company, for up to €230M in cash including a post-closing milestone component. Gleamer will be integrated into DeepHealth, RadNet's AI and technology subsidiary. Gleamer's platform covers AI-assisted detection across chest X-ray and thoracic CT workflows, and the deal extends DeepHealth's AI capabilities into a European-origin commercial product with existing global customer relationships.
No financial terms of the milestone structure were disclosed beyond the total envelope.
The deal is notable in the context of the broader medtech/digital health M&A wave around radiology AI: within the same week, Sectra acquired Oxipit (autonomous chest X-ray AI, Lithuania), and Siemens Healthineers expanded its distribution of Cortechs.ai's neuroimaging platform. Radiology AI is consolidating rapidly, with workflow automation, autonomous reading, and specialist imaging tools emerging as three distinct sub-segments attracting different buyer profiles. RadNet/DeepHealth's acquisition of Gleamer positions it in the middle tier: AI-assisted detection tools sold to radiology practices, rather than autonomous reading or infrastructure.
Take-Private: Select Medical Holdings / WCAS Consortium — $3.9B
Select Medical Holdings / Consortium (Ortenzio, Jackson, WCAS) — $3.9B take-private | March 2, 2026
Select Medical (NYSE: SEM), one of the largest U.S. operators of critical illness recovery hospitals and outpatient rehabilitation clinics, agreed to be taken private at $16.50 per share in cash, implying a $3.9B enterprise value. The buyer consortium is led by Executive Chairman and co-founder Robert A. Ortenzio and Senior EVP Martin F.
Jackson, alongside healthcare-focused PE firm Welsh, Carson, Anderson & Stowe. The offer represents an 18% premium to the company's unaffected share price from November 24, 2025, and a 25% premium to its 90-day volume-weighted average. Ortenzio and Jackson are rolling over equity rather than taking cash, aligning insider and PE incentives.
The transaction requires approval from unaffiliated shareholders, Hart-Scott-Rodino clearance, and other regulatory sign-offs, with closing expected mid-2026. The deal is not subject to a financing condition. Select Medical will be delisted from NYSE upon completion. Ortenzio and Jackson are rolling approximately 11.8% of shares. No go-shop provision — standard no-solicitation with fiduciary out. Company termination fee: approximately $66.5M; parent reverse termination fee: approximately $133M. Outside date: December 1, 2026, extendable to March 1, 2027.
Advisors: Goldman Sachs (financial advisor to Special Committee); Skadden, Arps, Slate, Meagher & Flom (legal counsel to Special Committee); Dechert LLP (legal counsel to Select Medical); Wells Fargo and J.P. Morgan (financial advisors to Consortium, also joint lead arrangers and bookrunners for committed debt financing); Cravath, Swaine & Moore (legal counsel to Consortium); Barclays (financial advisor to WCAS); Ropes & Gray (legal counsel to WCAS); Paul Hastings (legal counsel to debt financing sources).
Stake Buyout: Grünenthal / Kyowa Kirin International — EMEA Established Medicines JV Consolidation | Private Pharma | March 2, 2026
Grünenthal / Kyowa Kirin International | Minority Stake Buyout (JV Consolidation) | Established Medicines / Pain, Related | EMEA Commercial | March 2, 2026
| Parameter | Detail |
|---|---|
| Announced | March 2, 2026 |
| Deal value | Undisclosed |
| Structure | Grünenthal acquires Kyowa Kirin International's stake in a joint venture covering established medicines |
| Geography | EMEA (primary); Germany-led operations |
| Parties | Grünenthal GmbH (private, Aachen) acquires from Kyowa Kirin International plc (subsidiary of Kyowa Kirin Co., Ltd., Tokyo) |
Grünenthal, the privately held German pharmaceutical company best known for its pain portfolio, announced the acquisition of Kyowa Kirin International's stake in their joint venture covering established medicines across EMEA. Financial terms were not disclosed. The transaction consolidates Grünenthal's control over a commercial portfolio that had previously been managed jointly, removing a sharing partner from the economics of an established medicines franchise.
This deal is structurally representative of a category that rarely appears in Bloomberg or Dealogic deal tables: private-company JV consolidations involving established (off-patent or loss-of-exclusivity) pharmaceutical assets, with no disclosed consideration. The underlying commercial rationale is straightforward — established medicines JVs often outlive their strategic rationale as the originating companies' priorities diverge, and the party with stronger commercial infrastructure and geographic focus frequently buys out the other. Grünenthal's EMEA pain and established medicines franchise is among the largest of its kind held by a private European pharma company, making the consolidation of this JV consistent with long-term portfolio ownership strategy.
Acquisition: Intuitive Surgical / ab medica, Abex, Excelencia Robótica — ~€319M Southern European Distribution
Intuitive Surgical / ab medica, Abex, Excelencia Robótica | Acquisition | Surgical Robotics | Commercial | Completed March 1–2, 2026
| Parameter | Detail |
|---|---|
| Deal value | ~€319M |
| Completed | March 1–2, 2026 (originally announced January 2025) |
| Markets | Italy, Spain, Portugal, Malta, San Marino |
| Installed base transferred | 470+ da Vinci systems |
| Employees transferred | ~250 |
Intuitive Surgical completed the acquisition of its Southern European distribution operations from three regional distributors (ab medica in Italy, Abex in Spain/Portugal, Excelencia Robótica in Spain) covering more than 470 installed da Vinci systems across Italy, Spain, Portugal, Malta, and San Marino. The deal transitions Intuitive from third-party distribution to direct commercial operations across these markets. Originally announced January 2025, completion occurred on March 1–2, 2026. Approximately 250 employees transferred to Intuitive's direct organization.
Acquisition: Sectra / Oxipit — Autonomous Chest X-Ray AI | Digital Health / Radiology AI | March 2026
Sectra / Oxipit | Acquisition | Radiology AI (Autonomous Chest X-Ray) | Marketed | March 2026
Medical imaging IT company Sectra (STO: SECT B) agreed to acquire Lithuanian radiology AI developer Oxipit, with closing expected during March 2026. Oxipit holds the first CE Class IIb certification for autonomous AI in chest X-ray analysis — a regulatory distinction reflecting clinically validated autonomy rather than decision-support assistance. Its flagship ChestLink product automatically identifies high-confidence normal chest X-ray studies and removes them from the radiologist worklist, with 99.9% sensitivity and the capacity to automate approximately 40% of cases.
In 2026, Oxipit expanded into chest CT and musculoskeletal imaging. ChestLink was already available through Sectra's Amplifier Marketplace; the acquisition moves Sectra from distribution partner to direct owner of the underlying autonomous AI technology. No financial terms disclosed. The acquisition consolidates the radiology AI autonomy segment alongside the RadNet/Gleamer and Siemens/Cortechs.ai developments announced the same week.
Acquisition: BioSyent / Oral Science — $25.5M + Seller Royalty | Canadian Oral Health | Commercial
BioSyent / Oral Science | Acquisition | Oral Health (Dental Distribution) | Commercial | March 2, 2026
| Parameter | Detail |
|---|---|
| Purchase price at closing | $25.5M ($22.5M cash + 234,192 BioSyent common shares at $12.81/share = $3.0M) |
| RSU component | $0.2M of cash consideration settled via 12,666 RSUs to certain Oral Science employees (vest year 2) |
| Share escrow / lock-up | 167,635 shares in two-year escrow; 66,557 shares in two-year lock-up (25% released every six months) |
| Additional cash at closing | $2.0M (excess working capital above $6.3M NWC requirement) |
| Contingent earn-out | Cash payment in 2027 based on Oral Science 2025–2026 performance |
| Contingent royalties | Product-specific royalties through 2033, capped at $6.0M (single undisclosed product) |
| Total maximum consideration | ~$33.5M+ (closing + NWC + earn-out + royalty cap) |
| Acquirer | BioSyent Inc. (TSXV: RX), Mississauga, Ontario |
| Target | Oral Science Inc. (privately held; Brossard, Quebec; founded 2003) |
| Target TTM revenue | >$30M (standalone, TTM to Sept 30, 2025) |
| Funding | $16.3M cash on hand + $6.0M RBC 1-Year Senior Secured Demand Term Loan + $2.0M RBC revolving credit draw |
| Post-close net cash | >$10M retained (BioSyent) |
| Additional credit line | RBC Senior Secured Demand Revolving Credit Line up to $12.0M (combined with term loan not to exceed $12.0M) |
| Pro forma TTM revenue (combined) | >$70M |
| Pro forma TTM EBITDA (combined) | >$15.75M |
| Purchase multiple (closing) | 6.33x EBITDA (TTM Sept 30, 2025, before contingent consideration) |
| Founders / leadership | Oral Science founders and leadership team remain in place; become BioSyent shareholders |
| Exchange classification | TSXV Expedited Acquisition under Policy 5.3; subject to final TSXV approval |
| SPA signed | February 8, 2026 |
| Closed | March 2, 2026 |
BioSyent closed its acquisition of Oral Science Inc. on March 2, adding a profitable and growing Canadian dental products distributor to its pharmaceutical and oral health portfolio. Founded in 2003 and headquartered in Brossard, Quebec, Oral Science partners with dental practices across periodontal and endodontic disease, high-risk caries, dry mouth, and oral lesions. Its integrated approach is built on four pillars: diagnosis and monitoring, in-office treatments, home-care solutions, and clinical team and patient education. Revenue is split approximately 54% from dental clinics and the remainder through retail pharmacy and direct-to-consumer online channels.
Oral Science generated more than $30M in standalone trailing revenue for the twelve months ended September 30, 2025 — a business with scale, profitability, and established channel relationships. The founders and leadership team remain in place and become BioSyent shareholders through the share consideration, aligning their incentives with the combined company's performance. Oral Science continues operating from Brossard as a standalone business unit within BioSyent.
The royalty structure — why this deal is relevant to the royalty market
The contingent royalty consideration is the element of primary interest to pharmaceutical royalty participants. Under the SPA, Oral Science's former shareholders retain the right to receive product-specific royalties based on the future net sales of a single undisclosed product through 2033, capped at $6.0M in total. The specific royalty rate, the product name, and the base period are not disclosed — the $6.0M cap is the only economic boundary defined in the public filing.
This is a classic seller-retained royalty in a commercial asset acquisition: the acquirer takes ownership of the full operating business and bears commercialization risk and capital cost going forward, while the sellers retain a capped revenue-linked participation tied to the single most commercially sensitive product in the portfolio. The structure aligns the sellers' payout with the product's commercial trajectory rather than paying them a fixed earn-out that would be owed regardless of performance — a form of contingent consideration that is common in pharma and specialty healthcare M&A but less frequently seen in the dental distribution segment.
For royalty market participants, the structure raises a specific interpretive question: is the royalty tied to a branded, differentiated product with pricing power and a defensible market position — in which case the $6.0M cap may represent a meaningful floor rather than a ceiling — or to a commodity-adjacent product with limited growth and high substitution risk? The public filing does not identify the product, so this assessment must await BioSyent's next shareholder disclosure cycle. What is disclosed is that Oral Science carries proprietary products and exclusive distribution agreements alongside its branded partner portfolio, suggesting the royalty-bearing product may be on the proprietary or exclusively licensed end of the portfolio rather than a generic or white-label item.
BioSyent's capital position and deal rationale
The acquisition is funded predominantly from BioSyent's existing cash, drawing only $8.0M from the new RBC credit facilities (term loan + revolver draw), which management expects to fully retire by Q4 2026. Post-close, BioSyent retains more than $10M in net cash and investments alongside a $12.0M revolving line — a position that preserves meaningful capacity for further acquisitions or in-licensing within the coming twelve months. CEO René Goehrum described Oral Science as a "Canadian success story much like our own" and positioned the deal as delivering on BioSyent's strategic priorities of profitable growth, revenue diversification, and long-term value creation.
The combined pro forma EBITDA of more than $15.75M on more than $70M in revenue implies a blended margin of approximately 22.5%, which is healthy for a specialty healthcare distribution and marketing platform. At 6.33x EBITDA on the closing purchase price — before earn-out and royalties — the multiple is consistent with mid-market Canadian specialty healthcare acquisition comps and reflects the absence of late-stage development risk that typically commands higher multiples in biopharma M&A.
SPAC: VERAXA Biotech / Voyager Acquisition Corp. — Shareholder Approval | ADC/BiTE Oncology
VERAXA Biotech / Voyager Acquisition Corp. | SPAC Business Combination (Milestone) | ADC/BiTE Oncology | Clinical-Stage
| Parameter | Detail |
|---|---|
| VERAXA shareholder vote | 99.57% approval (12,680,593 votes at EGM, February 27, 2026) |
| Announced | March 2, 2026 |
| SPAC | Voyager Acquisition Corp. (Nasdaq: VACH); sponsored by Cantor Fitzgerald & Co., Voyager Acquisition Sponsor Holdco LLC, and Odeon Capital Group LLC |
| Target | VERAXA Biotech AG (Zurich; portfolio company of Xlife Sciences AG, SIX: XLS) |
| Transaction type | Absorption merger; Veraxa Biotech Holding AG acquires VERAXA Biotech AG and renames to Veraxa Biotech AG |
| Capital increase approved | Ordinary capital increase up to CHF 223,400 for share issuance to Voyager shareholders |
| Next gate | Voyager shareholder vote — scheduled March 12, 2026 |
| Post-close ticker | VRXA (NASDAQ) |
| Business combination agreement signed | April 22, 2025 |
| Proxy statement/prospectus filed | February 19, 2026 |
VERAXA Biotech AG, a Zurich-based oncology company focused on antibody-drug conjugates (ADCs), bispecific T cell engagers (BiTEs), and its proprietary BiTAC antibody format, cleared the final VERAXA-side prerequisite for its merger with Voyager Acquisition Corp. on February 27, 2026, with a 99.57% shareholder vote. VERAXA originates from scientific work conducted at the European Molecular Biology Laboratory (EMBL) and is developing a pipeline of next-generation antibody-based cancer therapies. The business combination agreement was originally signed in April 2025.
The March 2 announcement is a procedural milestone rather than a transaction closing — the Voyager shareholder vote remains the final gate, with trading under VRXA on NASDAQ expected to commence shortly after. VERAXA's management team, led by CEO Dr. Christoph Antz, continues unchanged. The transaction gives VERAXA access to public capital markets for clinical development of its ADC and BiTAC pipeline, consistent with the broader 2025–2026 wave of European oncology biotechs pursuing U.S. NASDAQ listings via SPAC as a route to institutional investor access.
Corporate Vote: BioMarin / Amicus Therapeutics — Merger Vote Completed March 3, 2026
BioMarin Pharmaceutical / Amicus Therapeutics | Acquisition | Rare Lysosomal Storage Disorders | Commercial | Vote March 3, 2026
| Parameter | Detail |
|---|---|
| Vote date | March 3, 2026 (virtual special meeting) |
| Deal terms | All-cash at $14.50/share (~$4.8B equity value); agreement dated December 19, 2025 |
| Shares eligible | 313,918,463 (record date January 28, 2026) |
| Shares voted | 234,785,243 (74.79% quorum) |
| Merger adoption vote | 234,593,492 FOR / 119,194 AGAINST / 72,557 abstentions (99.9%+ approval) |
| Executive compensation advisory vote | 209,150,012 FOR / 24,282,220 AGAINST (approved, non-binding) |
| HSR early termination | Granted February 11, 2026 |
| Remaining conditions | Regulatory clearances in certain European jurisdictions and Japan |
| Expected closing | Q2 2026 |
| Drop-dead date | December 19, 2026 |
| BioMarin financial advisors | Morgan Stanley, J.P. Morgan |
| BioMarin legal counsel | Jones Day (acquisition); Cooley LLP (financing) |
| Amicus financial advisors | Centerview Partners, Goldman Sachs (also provided fairness opinion) |
| Amicus legal counsel | Kirkland & Ellis |
| Debt financing | $3.7B package: $850M senior unsecured notes due 2034, $2B Term Loan B, $800M Term Loan A, $600M revolving credit facility |
| Leverage target | Gross leverage <2.5x within two years |
| Termination fee | $175M (Amicus to BioMarin) |
| Key assets acquired | Galafold (migalastat, Fabry disease; 2025 revenue $521.7M) + Pombiliti + Opfolda (Pompe disease; 2025 revenue $112.5M) |
| Pipeline acquired | DMX-200 (CCR2 inhibitor for FSGS, Phase 3 ACTION3 trial) |
| Royalty obligations inherited | Dimerix DMX-200 license: $30M upfront (paid) + up to $75M development/regulatory milestones + $35M first commercial sale milestone + up to $410M commercial milestones + up to $40M future-indication milestones = total potential ~$560M in milestones + low-teens to low-twenties % tiered royalties on U.S. net sales |
| Galafold generic entry | Teva, Aurobindo, Lupin generic license agreements permitting entry January 30, 2037 |
The Amicus Therapeutics special meeting on March 3, 2026 produced an overwhelming 99.9%+ shareholder vote in favor of BioMarin's all-cash acquisition at $14.50/share. The only remaining conditions to closing are regulatory clearances in select European jurisdictions and Japan; HSR (U.S. antitrust) clearance was obtained early in February. The transaction, first announced December 19, 2025, is expected to close in Q2 2026.
The deal carries a material inherited royalty obligation: BioMarin assumes the Dimerix license covering DMX-200, with up to approximately $560M in milestones plus low-teens to low-twenties percent tiered royalties on U.S. net sales. The acquisition also crystallizes the Galafold generic entry timeline — Teva, Aurobindo, and Lupin hold authorized generic agreements permitting market entry on January 30, 2037, providing BioMarin approximately 11 years of commercial runway on its largest acquired revenue stream.
Licensing
| # | Licensor | Licensee | Asset | Upfront | Total Potential | Royalty | Stage | Date |
|---|---|---|---|---|---|---|---|---|
| 1 | Chia Tai Tianqing (Sino Biopharma) | Sanofi | Rovadicitinib (JAK/ROCK) | $135M | Up to $1.53B | Up to double-digit tiered | Ph 3 / Approved (China) | March 4 |
| 2 | Antengene | UCB | ATG-201 (CD19/CD3 BiTE) | $60M + $20M near-term | Up to ~$1.18B | Tiered (undisclosed rate) | Ph 1 | March 3 |
| 3 | Pierre Fabre | Eton Pharmaceuticals | HEMANGEOL (propranolol) U.S. rights | $14M | $14M + royalties | 8% of U.S. net sales (patent life) | Marketed | March 2 |
| 4 | Celyn Therapeutics | Kairos Pharma | CL-273 (EGFR NSCLC) worldwide | None disclosed | $15M milestone + royalty | 2% of U.S. net revenues (IP life) | Pre-IND | March 2 |
| 5 | Orexo AB | Dexcel Pharma USA | Zubsolv (buprenorphine/naloxone) U.S. | ~$95M net | ~$111.8M total potential | Contingent consideration up to $16.8M on 2026–2027 sales | Marketed | March 3–4 |
| 6 | GAIA AG | Daiichi Sankyo Europe | Lipodia (digital therapeutic) Europe | Undisclosed | Undisclosed | Undisclosed | Approved (Germany DiGA) | March 5 |
| 7 | Helix | Alnylam | GenoSphere clinico-genomic cohort | Undisclosed | Undisclosed | N/A (data access) | Population genomics | March 5 |
| 8 | Nxera Pharma | Centessa Pharmaceuticals | ORX489 (OX2R agonist) milestone | — | $3M milestone triggered | Per collaboration agreement | Ph 1 | March 5 |
Section total (disclosed upfront): ~$209M upfront + $20M near-term = ~$229M immediately deployable. Total potential across disclosed deals: ~$2.74B+ (Sanofi $1.53B + UCB $1.18B + remainder). Orexo/Dexcel is an asset divestiture with royalty-adjacent contingent consideration.
Royalty notes: Sanofi/Chia Tai: "up to double-digit tiered" — precise floor not disclosed; publicly confirmed as double-digit but starting rate undisclosed. UCB/Antengene: tiered rate not disclosed in public filings; rate confirmation not available. Eton/Pierre Fabre: 8% explicitly disclosed — cleanest royalty rate in the section. Kairos/Celyn: 2% U.S. explicitly disclosed — low, consistent with pre-clinical asset range. Dexcel/Orexo: not a royalty per se but contingent consideration ($16.8M cap on 2026–2027 sales performance) functions as a seller-retained royalty-equivalent.
Research Collaboration: Alnylam / Tenaya Therapeutics — Up to $1.23B Cardiovascular RNAi Discovery Collaboration
Alnylam Pharmaceuticals / Tenaya Therapeutics | Research Collaboration + License | Cardiovascular Disease | Discovery | March 5, 2026
| Parameter | Detail |
|---|---|
| Announced | March 5, 2026 (agreement signed March 4) |
| Structure | Research collaboration + target-by-target exclusive license to Alnylam |
| Upfront payment | Up to $10M (payable within 30 days; subject to $500,000 reductions per target for up to 8 Tenaya-nominated targets failing agreed standards; theoretical minimum $6M) |
| Research cost reimbursement | Yes (from Alnylam to Tenaya) |
| Development and commercial milestones | Up to $1.13B |
| Royalties | Not disclosed; 8-K describes "development, regulatory and sales-based milestones" without mentioning traditional royalties — full terms expected as Tenaya Q1 2026 10-Q exhibit |
| Research structure | 24-month validation period (extendable) + 24-month evaluation period |
| Targets | Up to 15 novel genetic targets for cardiovascular disease (jointly nominated; deal is modality-agnostic, though Alnylam's core platform is RNAi) |
| Tenaya platform | iPSC-derived cardiomyocyte phenotypic screening |
| Downstream rights | Alnylam assumes all development, manufacturing, and commercialization per target; Tenaya has no co-development or co-commercialization rights |
| Termination right | Alnylam can terminate for any reason with notice |
| Advisors | No financial or legal advisors publicly disclosed for either party; consistent with research collaboration rather than M&A |
| Tenaya GC | Jennifer Drimmer Rokovich (signed Tenaya 8-K) |
| Headline figure note | "$1.23B" cited by some outlets appears to include ~$90M in estimated research reimbursement; Tenaya's 8-K cites $10M upfront plus up to $1.13B in milestones |
Alnylam Pharmaceuticals and Tenaya Therapeutics announced on March 5, 2026 a cardiovascular disease research collaboration in which Tenaya will deploy its iPSC-derived cardiomyocyte screening platform to validate up to 15 novel genetic targets over a 24-month research term. For each validated target, Alnylam receives an exclusive worldwide license and assumes all downstream development, manufacturing, and commercialization responsibilities.
The collaboration extends Alnylam's cardiovascular franchise — which already includes inclisiran (co-developed with Novartis), vutrisiran, fitusiran, and zilebesiran — into entirely new genetic target space identified through Tenaya's disease-relevant human cardiomyocyte models. The 24-month research horizon and milestone structure ($10M upfront, up to $1.13B in contingent payments) is consistent with Alnylam's established pattern of building its pipeline through platform access agreements before committing development capital. Tenaya retains no development responsibilities once targets are licensed to Alnylam.
Major Licensing: Sanofi / Chia Tai Tianqing (Sino Biopharmaceutical) — rovadicitinib | Up to $1.53B
Sanofi / Chia Tai Tianqing Pharmaceutical Group | Licensing | Myelofibrosis (China approval), Chronic Graft-versus-Host Disease (global Phase 3) | Phase 3 / Approved (China)
| Parameter | Detail |
|---|---|
| Announced | March 4, 2026 |
| Licensor | Chia Tai Tianqing Pharmaceutical Group Co., Ltd. (subsidiary of Sino Biopharmaceutical Holdings, HKEX: 1177) |
| Licensee | Sanofi |
| Rights granted | Exclusive worldwide license (development, manufacture, and commercialization) |
| Upfront payment | $135M |
| Development, regulatory, and sales milestones | Up to $1.395B |
| Total potential value | Up to $1.53B |
| Royalties | Up to double-digit tiered on annual net sales |
| Asset | Rovadicitinib (brand name: Anxu in China) — oral dual JAK/ROCK inhibitor |
| China approval | February 2026 (NMPA) — first-line treatment for intermediate-2 or high-risk PMF, PPV-MF, and PET-MF |
| Primary global target indication | Chronic graft-versus-host disease (cGVHD) |
| Phase 3 cGVHD status | NCT06682169, ongoing in China; FDA-cleared Phase 2 in the U.S. |
Announced March 4, 2026, this is the week's largest disclosed licensing transaction and the most prominent example of a China-to-West deal moving a fully approved domestic asset into a global development and commercialization program. Chia Tai Tianqing, the pharmaceutical flagship of Sino Biopharmaceutical — one of China's largest pharmaceutical groups by revenue — grants Sanofi worldwide exclusive rights to rovadicitinib, a first-in-class oral dual inhibitor of JAK1/2 (via the STAT3/5 pathway) and ROCK1/2 (via the anti-fibrotic axis). The dual mechanism is the scientific cornerstone of the asset's differentiation: most approved myelofibrosis therapies (ruxolitinib, fedratinib, pacritinib) are JAK inhibitors only, targeting the inflammatory cascade but not the fibrotic process that drives splenomegaly progression and bone marrow failure over time.
Rovadicitinib's simultaneous inhibition of ROCK1/2 adds an anti-fibrotic dimension theoretically capable of slowing the stromal remodeling underlying disease progression — a mechanistic distinction no current approved MF therapy can claim.
Rovadicitinib received NMPA approval in February 2026 under the brand name Anxu for first-line treatment of intermediate-2 or high-risk primary myelofibrosis (PMF), post-polycythemia vera MF (PPV-MF), and post-essential thrombocythemia MF (PET-MF) in adult patients — making it one of only a handful of novel MF therapies to achieve global regulatory approval. Sanofi's primary development target, however, is chronic graft-versus-host disease (cGVHD), a severe and often life-threatening complication of allogeneic hematopoietic stem cell transplantation where the JAK/STAT3/5 inflammatory and ROCK-mediated fibrotic pathways are both implicated in disease maintenance and organ damage. Sanofi's transplant and immunology franchise — which markets Enjaymo (sutimlimab) and previously commercialized Mozobil (plerixafor) — provides an established infrastructure into which rovadicitinib fits strategically.
The competitive cGVHD landscape includes ruxolitinib (Jakavi, Novartis/Incyte, FDA-approved 2021), ibrutinib (Imbruvica, J&J/AbbVie, FDA-approved 2017), and axatilimab (Niktimvo, Syndax/Incyte, FDA-approved 2024), along with several agents in later-stage development. Rovadicitinib's dual mechanism differentiates it on paper from all current approved agents in cGVHD, none of which address both inflammatory and fibrotic pathology simultaneously.
The deal economics are at the higher end of the China-to-West licensing spectrum for 2026. The $135M upfront is substantially larger than the $60M UCB paid to Antengene for ATG-201 (announced the same week), reflecting rovadicitinib's more advanced clinical status — already approved in China and in active Phase 3 in cGVHD. The up-to-double-digit tiered royalty structure, fully disclosed in public filings, places this among the more transparent royalty packages in recent China-originated out-licensing.
Competitive context in myelofibrosis: Rovadicitinib enters a global MF market anchored by ruxolitinib (Jakavi/Jakafi, Incyte/Novartis) and increasingly crowded with fedratinib (Inrebic, BMS), pacritinib (Vonjo, SOBI/CTI BioPharma), and momelotinib (Ojjaara, GSK). None of the existing approved agents have demonstrated meaningful anti-fibrotic activity at standard therapeutic doses. If Sanofi pursues an MF label expansion alongside cGVHD, rovadicitinib would compete directly in this market with a mechanistic claim no current approved competitor can make.
Major Licensing: UCB / Antengene — ATG-201
UCB / Antengene | Licensing | B Cell-Related Autoimmune Diseases | Phase 1
| Parameter | Detail |
|---|---|
| Upfront | $60M |
| Near-term milestones | $20M (on satisfaction of conditions) |
| Total upfront + near-term | $80M |
| Development/regulatory/commercial milestones | >$1.1B |
| Royalties | Tiered on future net sales |
| Equity component | None — pure license with cash payments and tiered royalties |
| Asset | ATG-201 (CD19/CD3 bispecific T-cell engager, AnTenGager™ platform) |
| CRS reduction mechanism | (1) Steric hindrance masking — cryo-EM confirmed CD3 binding site physically blocked until CD19 target engagement; (2) fast-on-fast-off CD3 binder scFv limiting activation duration; (3) bivalent 2+1 architecture (two CD19 arms, one CD3 arm) enhancing avidity while controlling T-cell engagement |
| Geography | Worldwide exclusive |
| Phase 1 execution | Antengene (China & Australia), then transfers to UCB |
| Advisors | No financial or legal advisors publicly disclosed; UCB on Euronext Brussels, Antengene on HKEX (6996) — neither SEC-registered |
Announced March 3, 2026, the UCB/Antengene deal is the week's most substantive licensing transaction. UCB gains worldwide exclusive rights to develop, manufacture, and commercialize ATG-201, a CD19/CD3 bispecific T-cell engager designed for B cell depletion in autoimmune diseases, along with access to Antengene's manufacturing technology and the underlying AnTenGager platform.
ATG-201 employs steric hindrance masking technology intended to reduce cytokine release syndrome risk — a key differentiator relative to first-generation T-cell engagers — along with a proprietary fast-on-fast-off CD3 binder designed to limit T-cell exhaustion. Preclinical data presented at ACR 2025 showed complete and sustained B cell depletion across blood, bone marrow, and spleen in humanized mouse models following a single dose.
The deal structure is characteristic of China-to-West licensing: Antengene retains responsibility for IND submissions in China and Australia in Q1 2026 and will conduct first-in-human Phase 1 studies before transferring further clinical development to UCB. The $80M upfront and near-term package and $1.1B+ milestone stack positions this among the larger China-originated autoimmune deals of early 2026.
Licensing: Eton Pharmaceuticals / Pierre Fabre — HEMANGEOL U.S. Commercialization Rights | $14M Upfront + Royalty
Eton Pharmaceuticals / Pierre Fabre | Licensing | Infantile Hemangioma | Marketed (FDA Orphan Drug)
| Parameter | Detail |
|---|---|
| Upfront payment | $14.0M |
| Royalty | 8% of U.S. net sales for patent life |
| Inventory at closing | ~$1.5M |
| Additional inventory (May 2026) | ~$0.7M est. |
| Total initial outlay | ~$16.2M |
| U.S. commercialization start | May 1, 2026 |
| Licensor | Pierre Fabre Medicament Sas (continues global ex-U.S. rights) |
| Licensee | Eton Pharmaceuticals, Inc. (Nasdaq: ETON) |
| Financing | Cash on hand; expected accretive to 2026 earnings |
| Agreement signed | February 27, 2026 |
| Announced | March 2, 2026 |
Eton Pharmaceuticals announced on March 2 the in-licensing of U.S. commercialization rights to HEMANGEOL (propranolol hydrochloride) oral solution from Pierre Fabre, the only FDA-approved systemic therapy for proliferating infantile hemangioma. The asset is a marketed Orphan Drug treating an estimated 5,000–10,000 U.S. infants annually. Pierre Fabre retains global commercialization rights excluding the U.S. and will continue U.S. distribution through April 30, 2026.
The deal gives Eton its tenth commercial rare disease product — reaching a stated long-held milestone — and fits squarely within its orphan drug-focused commercial model. Eton will deploy its Eton Cares patient support program, which includes a $0 commercial co-pay for qualifying patients, specialty distribution, benefits investigation, and patient education services tailored to the pediatric workflow.
The royalty structure (8% of U.S. net sales for patent life) is a textbook ex-U.S. originator royalty arrangement: Pierre Fabre exits U.S. commercial operations while retaining a recurring revenue stream on the market it built. For Eton, the deal converts a one-time outlay of roughly $16M into a royalty-bearing commercial asset expected to generate immediate earnings accretion, financed from existing cash.
Licensing: Kairos Pharma / Celyn Therapeutics — CL-273 Exclusive Worldwide Rights | Equity + Milestone
Kairos Pharma / Celyn Therapeutics | Asset Acquisition (Binding Terms) | EGFR-Mutant NSCLC | Pre-IND
| Parameter | Detail |
|---|---|
| Asset | CL-273 — AI-designed, reversible, wild-type-sparing pan-EGFR small-molecule inhibitor |
| Seller | Celyn Therapeutics, Inc. (privately held; backed by OrbiMed and Torrey Pines Investment) |
| Acquirer | Kairos Pharma, Ltd. (NYSE American: KAPA) |
| Equity consideration | 16.5% of Kairos on fully diluted basis at closing |
| Regulatory milestone | $15M (cash + shares mix) upon FDA NDA/BLA submission |
| Royalty | 2% of U.S. net revenues for IP life |
| Additional asset included | CL-741, a Phase 1-ready oral type IIb c-MET inhibitor |
| Upfront cash | None disclosed |
| Conditions | Shareholder approvals (both parties); no material adverse effect; NYSE American clearance if required |
| Financial advisor | D. Boral Capital, LLC (sole advisor to Kairos) |
| Announced | March 2, 2026 |
Kairos Pharma signed binding terms on March 2 to acquire exclusive worldwide rights — covering development, manufacturing, commercialization, and all related IP and regulatory rights — to CL-273 from Celyn Therapeutics. The deal also includes CL-741, a Phase 1-ready c-MET inhibitor, providing Kairos with a complementary asset targeting MET-driven resistance in NSCLC.
CL-273 is an AI-discovered, pre-IND small-molecule designed to block multiple EGFR mutant forms (including C797S resistance mutations that arise following osimertinib treatment) while sparing wild-type EGFR — a profile intended to improve the therapeutic index over first- and third-generation EGFR inhibitors. The compound completed GLP toxicology studies and first-in-human trials are projected for 2026. EGFR mutations are present in roughly 10–15% of NSCLC cases in Western populations and up to 50% in East Asian populations; the addressable market is estimated at $16.2B in 2026.
The consideration structure — equity-only at closing, a single regulatory milestone, and a low royalty rate — reflects Kairos's limited cash position (market cap ~$0.63/share at announcement) and Celyn's willingness to accept paper and future contingencies in exchange for development momentum. The 2% U.S. royalty retained by Celyn/OrbiMed is consistent with pre-clinical asset royalty ranges and reflects the early stage of the asset.
Asset Divestiture & Debt Elimination: Dexcel Pharma USA / Orexo AB — Zubsolv® + Bond Redemption
Dexcel Pharma USA / Orexo AB | Asset Purchase + Debt Elimination | Opioid Use Disorder | Marketed
Note: The Zubsolv asset purchase closed January 7, 2026 (signed December 22, 2025), outside the W10 reporting window. The W10 event is the bond redemption call announced March 4, 2026, which completes the two-step strategic sequence.
| Parameter | Detail |
|---|---|
| Structure | Asset Purchase Agreement (full U.S. rights + commercial infrastructure) |
| Fixed purchase price | $91M cash at closing |
| Inventory payment | $3.8M at closing |
| Contingent consideration | Up to $16.8M based on Zubsolv net sales 2026–2027 |
| Total potential consideration | ~$111.8M |
| Zubsolv LTM net sales at signing | ~$49M |
| Zubsolv LTM EBIT at signing | ~$17M |
| U.S. commercial employees | Majority transferred to Dexcel Pharma USA |
| Legal counsel (buyer) | DLA Piper LLP (US) |
| Asset purchase closed | January 7, 2026 |
| Bond: instrument | SEK 500M senior secured social bonds 2024/2028 (ISIN: SE0021515277) |
| Bond: redemption price | 103.25% of nominal amount + accrued interest |
| Bond: record date | March 23, 2026 (registered holders in Euroclear Sweden) |
| Bond: settlement date | March 30, 2026 |
| Bond: delisting | Nasdaq Stockholm sustainable bonds list, concurrent with settlement |
| Bond redemption announced | March 4, 2026 |
| Post-redemption debt position | Zero (fully debt-free) |
The Dexcel Pharma / Orexo transaction is a two-act strategic sequence that concludes in W10. Act one — the January close of the Zubsolv U.S. asset sale for $91M cash — was the instrument. Act two — the March 4 early redemption call on the SEK 500M bond — is the execution of the stated purpose.
Orexo's SEK 500M senior secured social bonds (issued 2024, maturity 2028) carried a call premium of 3.25% above nominal. At current SEK/USD rates, the total redemption outlay is approximately $46–47M equivalent, meaning the $91M cash from Dexcel more than covers the full bond retirement including the premium and accrued interest, while leaving Orexo with residual cash to fund its pipeline. Registered bondholders in Euroclear Sweden's debt register as of March 23, 2026 will receive the Redemption Price on March 30, 2026. The bonds will simultaneously be delisted from Nasdaq Stockholm's sustainable bonds list — the ESG-labelled debt structure that Orexo had used to market the 2024 issuance to socially conscious fixed income investors.
What the bond was and why it mattered
The 2024/2028 social bond was issued at a time when Orexo still held Zubsolv and needed balance sheet flexibility. Structuring it as a "social bond" — tied to the therapeutic impact of Zubsolv in opioid use disorder treatment — allowed Orexo to access the ESG-labelled debt market, which generally prices at a modest premium to conventional bonds for qualifying issuers. The irony is that the divestiture of Zubsolv to Dexcel removed the very asset underpinning the social bond's ESG rationale, making redemption not just financially logical but practically necessary: continuing to carry a social bond labelled against an asset you no longer own would be a governance and disclosure liability.
Post-divestiture Orexo: what the cash enables
Debt-free and with residual Zubsolv sale proceeds in hand, Orexo's capital allocation now points entirely to its AmorphOX platform pipeline. The three active programs are Izipry (naloxone nasal spray for opioid overdose, approved), OX640 (epinephrine intranasal for anaphylaxis, Phase 2), and OX390 (novel NCE intranasal for adulterated opioid overdoses involving xylazine and medetomidine, BARDA-partnered at up to $50.9M, preclinical/IND-enabling stage). The contingent consideration of up to $16.8M tied to Zubsolv 2026–2027 net sales under Dexcel's stewardship represents a further cash inflow that Orexo has effectively converted from operational risk into a passive receivable — a residual royalty-like stream on a business it no longer manages.
Licensing: Daiichi Sankyo Europe / GAIA AG — Lipodia Digital Therapeutic (European Exclusive)
Daiichi Sankyo Europe / GAIA AG | Licensing | Hypercholesterolaemia | Approved (Germany DiGA) | March 5, 2026
Daiichi Sankyo Europe obtained exclusive European commercialization rights to Lipodia, a digital therapeutic developed by Hamburg-based GAIA AG for adults with hypercholesterolaemia. Germany is the initial market via the DiGA (Digitale Gesundheitsanwendungen) statutory reimbursement pathway, with expansion planned to other major European markets. The Phase 3 DIGICHOL trial is in final stages. This represents Daiichi Sankyo's first digital therapeutic in cardiovascular care. Financial terms undisclosed.
Data Access: Alnylam / Helix — GenoSphere Clinico-Genomic Cohort Access
Alnylam Pharmaceuticals / Helix | Data Access Agreement | RNAi Target Discovery | Population Genomics | March 5, 2026
Alnylam gained multi-year access to Helix's GenoSphere clinico-genomic cohorts — Exome+ sequencing data linked to longitudinal clinical records with an average 13-year EHR history — to identify and validate new RNAi therapeutic targets. The collaboration leverages population-scale genomic data for target discovery support across Alnylam's pipeline programs. Financial terms undisclosed.
Milestone: Nxera Pharma / Centessa Pharmaceuticals — $3M ORX489 Development Milestone
Nxera Pharma / Centessa Pharmaceuticals | Milestone Payment | Narcolepsy | Phase 1 | March 5, 2026
Centessa Pharmaceuticals achieved an early development milestone payment of $3M for ORX489, an oral orexin receptor 2 agonist for narcolepsy, under their existing research collaboration with Nxera Pharma (formerly Heptares). This follows a $1.8M milestone paid in February 2026, bringing total milestone receipts to date to $4.8M.
Royalty Financing and Non-Dilutive Capital
| # | Royalty Fund | Counterparty | Asset / Product | Amount | Structure | Royalty % | Cap / Return | Date |
|---|---|---|---|---|---|---|---|---|
| 1 | Royalty Pharma | Zymeworks (Ziihera/zanidatamab) | Ziihera (HER2 bispecific) | $250M | Royalty-backed note | 30% of worldwide tiered royalties from Jazz/BeOne | 1.65x by Dec 31 2033; 1.925x thereafter | March 2 |
| 2 | HCRx + Soleus Capital | Heidelberg Pharma | TLX250-Px royalties from Telix | $45M ($20M now + $25M FDA approval) | Amendment to royalty purchase; Soleus added as new participant | Double-digit (Heidelberg retains); royalty sold portion undisclosed | Increased cap; high single-digit tail after cap reverts to Heidelberg | March 7 |
| 3 | DRI Healthcare | Portfolio / capital markets | 29 royalty streams on 23 products | $330M+ (refinancing: C$108.7M debentures + $250M senior notes) | Capital markets refinancing; no new royalty acquisitions | N/A — balance sheet transaction | 5-yr target $800M–$1B deployment | March 2–3 |
| 4 | Athyrium + HCRx | Esperion (Japan bempedoic acid royalty) | Otsuka Japan royalties on NEXLETOL/NEXLIZET | Undisclosed (proceeds fund $75M Corstasis acquisition) | Royalty monetization (purchase agreement) | 15–30% underlying from Otsuka; portion sold to Athyrium/HCRx | Undisclosed cap and return threshold | March 3 |
Section total new capital deployed: ~$295M+ in new royalty financing (Royalty Pharma $250M + Heidelberg $20M now + Athyrium/HCRx undisclosed); $330M+ DRI refinancing (balance sheet, not new deployment).
Royalty notes — gaps: Zymeworks/Royalty Pharma: the "worldwide tiered royalties" rate that Zymeworks earns from Jazz and BeOne on Ziihera net sales is not publicly disclosed — only that Royalty Pharma receives 30% of that undisclosed underlying rate. Heidelberg/HCRx: the proportion of Heidelberg's double-digit royalty sold to HCRx/Soleus is not disclosed. Athyrium/HCRx/Esperion: deal size (lump sum received by Esperion), return threshold, and term are all undisclosed — only the underlying Otsuka rate (15–30% tiered) is public.
DRI Healthcare, Heidelberg / HCRx, and Royalty Pharma — Market Scale | March 2–7, 2026
DRI Healthcare — $330M+ Capital Markets Transactions | March 2–3, 2026
DRI Healthcare Trust executed two capital market transactions in consecutive days, collectively representing the most active single-week refinancing by a dedicated pharmaceutical royalty fund in 2026 to date.
On March 2, DRI entered subscription agreements with EdgePoint Wealth Management and Alberta Investment Management Corporation (AIMCo) to exchange existing preferred securities for C$108.7M (~$79M) in convertible debentures at 5.75%, maturing 2031 (conversion price C$21.99/unit; exclusive placement agent: Scotia Capital Inc.; non-redeemable before February 28, 2029; US$35.6M of preferred securities remain outstanding post-exchange). On March 3, DRI priced $250M in senior secured notes in two tranches ($106M at 5.35% due 2031 and $144M at 5.65% due 2033), with proceeds allocated to repaying existing acquisition facilities and funding portfolio growth. Combined with separately reported record FY2025 results — total income of $198.6M, up 6% year-over-year — DRI simultaneously announced a new five-year deployment target of $800M–$1B against a pipeline it characterizes at approximately $3B.
CEO Ali Hedayat confirmed DRI achieved $1.25 billion in total committed deployments over the prior five-year period, exceeding its original $850M–$900M target. The new 2026–2030 target is described as "fully funded" with no additional equity required, supporting a low-teens CAGR in adjusted EBITDA through 2030. The pipeline of ~$3B is skewed toward pre-commercial assets overall, though nearer-term opportunities (6–8 month horizon) are weighted toward post-approval drugs. The portfolio currently encompasses 29 royalty streams on 23 products including Ekterly, Eylea, Keytruda, Orserdu, Spinraza, Stelara, Casgevy, Xolair, and others. No new royalty acquisitions were announced during the March 2–7 window.
The back-to-back transactions reflect a structural dynamic becoming more common among mid-tier royalty funds: using low-cost fixed-rate debt to pre-fund future royalty acquisitions rather than drawing on revolving credit at close. At 5.35–5.75%, the notes are attractively priced relative to the unlevered yield profile of royalty portfolios, creating positive carry on deployed capital.
Heidelberg Pharma / HealthCare Royalty (HCRx) — Royalty Amendment + Soleus Capital Addition | March 7, 2026
Heidelberg Pharma amended its royalty financing agreement with HealthCare Royalty on March 7 — the second such amendment to the original March 2024 deal — adding Soleus Capital (acting via Soleus Capital Credit Opportunities Fund I) as a new participant. Historical deal evolution: the original March 2024 agreement provided $25M upfront + up to $75M on FDA approval + $15M sales milestone (total ~$115M); a first amendment in March 2025 added $20M and adjusted the cap structure; the March 2026 second amendment adds Soleus Capital's $45M total commitment ($20M upfront + $25M upon FDA approval). The amended terms provide Heidelberg with $20M upfront plus $25M upon FDA approval for royalties on Telix Pharmaceuticals' TLX250-Px imaging agent (Zircaix™) — a radiolabeled girentuximab targeting CAIX on clear cell renal carcinomas. Heidelberg retains double-digit royalties on TLX250-Px net sales from Telix, having originally licensed the asset to Telix in 2017. The amended structure includes an increased cap on total cumulative royalties sold and a high single-digit royalty tail (up from low single-digit in the original deal) payable after the cap is reached, after which remaining royalties revert to Heidelberg. The combined facility secures Heidelberg's financing through mid-2027.
Heidelberg ADC pipeline context. Heidelberg uses its proprietary ATAC® technology (amanitin-based ADCs). HDP-101 (BCMA ATAC for multiple myeloma; Orphan Drug + Fast Track designations) is the lead clinical candidate. HDP-102 (CD37 ATAC for NHL) is in clinical development; HDP-103 (mCRPC) and HDP-104 (GI tumors) have completed preclinical development. The announcement was made via EQS ad-hoc disclosure under EU Market Abuse Regulation Article 17 MAR (Ref: 2287470).
The Soleus addition is structurally notable: it converts a bilateral royalty purchase into a syndicated facility, with HCRx and Soleus holding proportional interests. Syndication of royalty facilities remains less common than in conventional credit markets but has been adopted with increasing frequency for assets where the underlying royalty payment timeline is uncertain (here, contingent on FDA approval). The amendment gives Heidelberg access to the full $45M committed facility while distributing funder risk.
Royalty Pharma — Asia Expansion Signal + Zymeworks $250M Deal | March 2, 2026
Royalty Pharma appointed Kenneth Sun as Senior Vice President and Head of Asia on March 2, the company's first dedicated Asia-focused senior hire. Sun joins from Morgan Stanley, where he was Managing Director and Head of Asia Pacific Healthcare Investment Banking, with prior roles at China Renaissance and BNP Paribas across 18+ years in Greater China healthcare banking. He will be based in Hong Kong. CEO Pablo Legorreta framed the hire around a $130 billion out-licensing market from Chinese medicines in 2025 (up from approximately $14B in 2021); Royalty Pharma aims to build the royalty market in Greater Asia analogous to its foundational role in establishing the Western biopharma royalty market. The appointment follows Gibson Dunn's 2026 royalty market outlook, which reported aggregate royalty transactions reached a record ~$6.5B in 2025, with 87% of surveyed biopharma executives indicating plans to pursue royalty financing within three years. Royalty Pharma's formal Asia build-out — Japan, South Korea, and Greater China are the primary target markets — reflects both the geographic expansion of royalty-eligible assets (particularly Japanese NHI-approved products generating domestic royalty streams) and the increasing sophistication of Asian biopharma executives about non-dilutive capital structures.
Announced the same day, Royalty Pharma also entered into a $250M royalty-backed note financing with Zymeworks (NYSE: ZYME) for Ziihera® (zanidatamab), a bispecific HER2-targeting antibody. Under the agreement, Royalty Pharma provides $250M and will be repaid from 30% of worldwide tiered royalties owed to Zymeworks from Jazz Pharmaceuticals and BeOne Medicines, capped at 1.65x by December 31, 2033 (or 1.925x if paid after that date). Zymeworks retains 70% of the royalties flowing from its licensing partners and all remaining regulatory and commercial milestone payments (up to $1.5B disclosed remaining). The structure is a royalty-backed note — a lending instrument repaid from royalty flows — rather than a royalty purchase, preserving Zymeworks' residual royalty participation while providing $250M of immediate non-dilutive capital. BeOne Medicines is an Asian-based licensing partner, making this deal directly relevant to Royalty Pharma's concurrent Asia expansion strategy.
Debt and Structured Financing
| # | Issuer | Counterparty / Trustee | Amount | Type | Key Terms | Date |
|---|---|---|---|---|---|---|
| 1 | AbbVie | U.S. Bank Trust / syndicate banks | $7.95B (7 tranches, 2028–2066) | Senior unsecured notes | Repays $4B 364-day term loan; maturities 2028–2066; rates 3.775%–5.65% fixed + SOFR floating | March 4 |
| 2 | UroGen Pharma | Pharmakon Advisors (BioPharma Credit) | $200M | Senior secured term loan | Royalty-adjacent: repays prior facility; secured on UroGen assets including Jelmyto royalty receivables | W10 |
Section total: ~$8.15B. AbbVie dominates; UroGen is the only biopharmaceutical-sector structured debt deal with royalty implications.
Royalty notes: UroGen's $200M term loan with BioPharma Credit (a healthcare-specialist lender) is secured against UroGen's commercial assets including Jelmyto (mitomycin) revenue streams — review section entry for full collateral detail.
Debt Issuance: AbbVie — $7.95B Multi-Tranche Senior Notes Offering | March 4, 2026
AbbVie Inc. | Senior Notes Offering | Investment Grade Corporate Debt | March 4, 2026
| Parameter | Detail |
|---|---|
| Total proceeds | ~$7.95B net proceeds (announced as $7.5B aggregate principal; upsized at pricing) |
| Closing date | March 4, 2026 |
| Purpose | Repay $4.0B 364-day delayed draw term loan maturing May 2026; general corporate purposes including potential debt repurchase |
| Tranche 1 | $750M floating rate notes due 2028 (Compounded SOFR-linked) |
| Tranche 2 | $1.5B 3.775% senior notes due 2028 |
| Tranche 3 | $1.25B 4.125% senior notes due 2031 |
| Tranche 4 | $1.25B 4.400% senior notes due 2033 |
| Tranche 5 | $1.5B 4.750% senior notes due 2036 |
| Tranche 6 | $1.25B 5.550% senior notes due 2056 |
| Tranche 7 | $500M 5.650% senior notes due 2066 |
| Security status | Unsecured, unsubordinated; rank pari passu with all existing senior unsecured debt |
| Registration | Registered under Securities Act; shelf registration Form S-3ASR (Feb 14, 2025) |
| Governing indenture | Base Indenture dated November 8, 2012, supplemented by Supplemental Indenture No. 12 dated March 4, 2026 |
| Trustee | U.S. Bank Trust Company, National Association |
| Underwriting agreement | February 24, 2026; syndicate of major banks |
| Covenants | Customary: limits on liens securing funded indebtedness; restrictions on consolidation/merger/asset transfer |
| Issuer credit | AbbVie Inc. (NYSE: ABBV); ~$360B market cap; FY2025 revenue ~$56B |
AbbVie entered into an underwriting agreement on February 24, 2026 and closed its largest-ever single bond offering on March 4, raising approximately $7.95B across seven tranches with maturities from 2028 to 2066. The immediate use of proceeds is repayment of a $4.0B 364-day delayed draw term loan originally drawn to support its pipeline and commercial activities; excess proceeds extend the company's liquidity and reduce shorter-dated debt. The maturity ladder — ranging from two-year floating rate paper to a 40-year 5.65% 2066 note — reflects AbbVie's standard practice of managing refinancing risk by distributing maturities across cycles.
The offering is notable for its scale and context. AbbVie entered 2026 with significant near-term maturities following the Allergan acquisition debt that has been progressively refinanced since 2020. The $7.95B raise is the single largest bond transaction in the pharmaceutical sector in W10 2026. The inclusion of 30- and 40-year paper at 5.55% and 5.65% — while higher than AbbVie's historical all-in cost of debt — reflects current risk-free rates and a modest investment-grade spread, consistent with AbbVie's A-range credit profile. The floating-rate 2028 tranche (SOFR-linked) provides optionality if rates decline before the 364-day term loan matures in May 2026.
From a capital structure standpoint, the transaction is leverage-neutral but maturity-extending: the company is replacing a short-dated loan with long-dated bonds. AbbVie's FY2026 guidance of $14.37–14.57 EPS implies sufficient free cash flow to service this new debt stack without altering shareholder return programs. The notes were issued under an automatic shelf registration, consistent with AbbVie's practice of maintaining a continuously effective S-3 to access capital markets at short notice.
Debt Refinancing: UroGen Pharma / Pharmakon Advisors (BioPharma Credit) — $200M Term Loan
UroGen Pharma / Pharmakon Advisors | Debt Refinancing | Urothelial Cancer | Commercial | March 2, 2026
| Parameter | Detail |
|---|---|
| Announced | March 2, 2026 |
| Facility | $200M refinanced term loan |
| Interest rate | 8.25% fixed |
| Amortization extended to | Q1 2030 |
| Lender | BioPharma Credit (Pharmakon Advisors) |
UroGen Pharma refinanced its existing term loan facility with BioPharma Credit (managed by Pharmakon Advisors) on March 2, 2026. The $200M facility was refinanced at 8.25% fixed interest with amortization extended to Q1 2030, described by management as reducing the company's cost of capital meaningfully. UroGen is a commercial-stage specialty urothelial cancer company with marketed products in bladder and upper tract urothelial cancers. BioPharma Credit is one of the primary dedicated life sciences lending platforms, with a portfolio of senior secured loans to commercial-stage biopharma companies.
Equity Financings and Investments
| # | Company | Round / Type | Amount | Lead Investors | Indication / Focus | Stage | Date |
|---|---|---|---|---|---|---|---|
| 1 | Generate:Biomedicines | IPO (Nasdaq: GENB) | ~$425M (incl. overallotment) | Flagship Pioneering; Novartis, Samsung, NVIDIA strategic | AI drug discovery / severe asthma | Ph 3-bound | Closed March 2 |
| 2 | Cognito Therapeutics | Series C | $105M (oversubscribed) | Morningside Ventures, IAG Capital, Starbloom Capital | Alzheimer's disease (Spectris neurostimulation) | Pivotal | March 5 |
| 3 | Science Corp | Series C | $230M | Undisclosed | Neurotech / BCI (Science Eye visual prosthesis) | Clinical | March 5 |
| 4 | Atavistik Bio | Series B extension | $40M | RA Capital | AKT1 inhibitor (hereditary hemorrhagic telangiectasia) + JAK2 (MPN) | Clinical | March 5 |
| 5 | NexCure | Private round | $19M | RA Capital, Cencora Ventures, Oncology Ventures | Outpatient CAR-T / T-cell engager treatment centers | Commercial build | W10 |
| 6 | MACC Fund / Versiti | Foundation investment | $11.5M | MACC Fund | Oncology / blood health research | Research | W10 |
| 7 | The Mark Foundation | Drug discovery grants | $4M (4 × $1M) | The Mark Foundation | Oncology academic translational | Early discovery | March 3 |
| 8 | Alithea Genomics | Seed (add-on close) | CHF 6.9M (~$7.8M) | Undisclosed | RNA transcriptomics tools for drug screening | Seed | March 4 |
| 9 | Shuttle Pharmaceuticals | Public offering | ~$3.5M | E.F. Hutton (placement agent) | Oncology / radiation sensitization | Clinical | March 6 (close March 9) |
| 10 | Rakovina Therapeutics | Private placement | C$1M | Undisclosed | Oncology / antiviral DNA damage | Clinical | W10 |
Section total (disclosed): ~$838M USD equivalent. Generate:Biomedicines ($425M IPO) and Science Corp ($230M) account for $655M. Cognito adds $105M. Remainder $48M across smaller rounds.
IPO: Generate:Biomedicines — $400M Nasdaq IPO (Offering Closed March 2, 2026)
Generate:Biomedicines | IPO | AI Drug Discovery / Severe Asthma | Phase 3-bound | Closed March 2, 2026
| Parameter | Detail |
|---|---|
| IPO size | $400M priced; grew to ~$425M after partial overallotment exercise |
| Share price | $16/share (midpoint of $15–$17 range) |
| Pricing date | February 26, 2026 |
| First trading date | February 27, 2026 |
| Offering close | March 2, 2026 |
| Ticker | Nasdaq: GENB |
| Designation | Largest biotech IPO of 2026 to date |
| Flagship Pioneering purchase | $75M at closing |
| Strategic investors | Novartis, Samsung, NVIDIA |
| Joint lead book-runners | Goldman Sachs, Morgan Stanley |
| Book-running managers | Piper Sandler, Guggenheim Securities, Cantor Fitzgerald |
| Company legal counsel | Goodwin Procter LLP |
| Underwriter legal counsel | Latham & Watkins LLP |
| Lock-up | 180 days (ending approximately August 26, 2026) |
| Lead asset | GB-0895 (anti-TSLP antibody) — two Phase 3 trials (SOLAIRIA-1/2) for severe asthma |
| Use of proceeds | ~$300M: SOLAIRIA-1/2 Phase 3 (severe asthma); ~$100M: GB-0895 COPD development; ~$75M: platform/technology/new programs; ~$15M: GB-4362 and GB-5267 Phase 1 oncology; remainder: general corporate |
| Licensing obligations | Amgen collaboration (2022): up to $1.9B across 6 programs; Novartis collaboration (2024): >$1B; Generate receives ~$31.9M in R&D collaboration revenue (2025) |
Note: Priced February 26–28, 2026; offering formally closed March 2. Included in the W10 window on the basis of the closing date.
Generate:Biomedicines, the Flagship Pioneering-backed AI drug developer, completed the largest biotech IPO of 2026 with a $400M offering closing March 2. The company's lead asset GB-0895 is an anti-TSLP antibody for severe asthma entering Phase 3, competing in a class anchored by AstraZeneca's tezepelumab (Tezspire). The company's AI-first drug design platform — trained across protein, nucleotide, and small molecule modalities — is its core differentiator. NVIDIA's participation as a strategic investor reflects the GPU-intensive compute demands of Generate's generative AI approach to protein design.
Small-Cap Public Offering: Shuttle Pharmaceuticals — ~$3.5M Public Offering
Shuttle Pharmaceuticals | Public Offering | Oncology / Radiation Sensitization | Clinical | March 6, 2026
Shuttle Pharmaceuticals announced a public offering of 2,238,800 common shares and pre-funded warrants for 4,761,200 shares under an S-1 registration statement, with E.F. Hutton acting as exclusive placement agent. Expected gross proceeds approximately $3.5M. Offering expected to close March 9, 2026.
Private Placement: Rakovina Therapeutics — C$1M Convertible Debenture
Rakovina Therapeutics | Private Placement | Oncology | Clinical | Closed March 5–6, 2026
Rakovina Therapeutics completed a non-brokered private placement of C$1M in convertible debenture units at C$50,000 each, with attached warrants (exercise price C$0.20, expiry January 2029). The company also settled C$1.59M in outstanding debt through share issuances in the same period.
Oncology & Cell Therapy
RA Capital Management, Cencora Ventures, Oncology Ventures / NexCure, Inc. | Equity Investment | Cell Therapy
$19M equity investment into NexCure, a company building and operating outpatient CAR-T and T-cell engager treatment centers. RA Capital Management is the company builder and lead investor; Cencora Ventures and Oncology Ventures participated as co-investors. Cencora Ventures' involvement is strategically notable — Cencora (formerly AmerisourceBergen) has been selectively building a direct investment portfolio in specialty therapeutics, and cell therapy distribution and reimbursement infrastructure is a logical focus given its specialty pharmacy and oncology distribution networks.
NexCure's model addresses a structural bottleneck: only approximately 150 of 6,100 U.S. hospitals currently administer CAR-T therapy.
MACC Fund / Versiti Blood Research Institute | Equity Investment | Oncology
$11.5M investment. MACC Fund is a Milwaukee-based children's cancer foundation; Versiti is a blood health research and diagnostics organization. The investment supports oncology research at the interface of blood health and pediatric/adult cancer biology.
The Mark Foundation for Cancer Research — 2026 Drug Discovery Awards | Philanthropy / Grants | Oncology (Multiple)
$4M total ($1M per award) distributed across four academic research teams. Announced March 3, 2026. The Drug Discovery Award is The Mark Foundation's translational grant program designed to bridge early academic discovery and clinical development — funding rigorous lead identification and optimization work at a stage that federal agencies find too applied and venture capital finds too early.
Grantees receive funding, CRO guidance, industry advisor access, and commercialization support through the Foundation's venture arm.
The four 2026 recipients and programs:
| Award | Investigators | Institution | Program |
|---|---|---|---|
| Drug Discovery Award | Aashish Manglik MD PhD, Michelle Arkin PhD, Boris Bastian MD PhD | UCSF | Directly Targeting Gq/11 Proteins for Uveal Melanoma Therapy — first direct inhibitors of the primary genetic drivers of uveal melanoma, a rare lethal eye cancer |
| Drug Discovery Award | Steven Corsello MD, Nathanael Gray PhD | Stanford University | TRIM21 E3 Ligase Reprogramming for Cancer Therapy — first-in-class targeted protein degradation approach for pancreatic cancer |
| Drug Discovery Award | William Sellers MD, Alex Burgin PhD | Broad Institute of MIT and Harvard | Biparatopic Antibodies Targeting Receptor Tyrosine Kinases in Cancer — high-precision biparatopic antibodies for aggressive solid tumors combining growth inhibition and drug delivery |
| Drug Discovery Award | Eric Fischer PhD, Scott Armstrong MD PhD | Dana-Farber Cancer Institute | A Novel Molecular Glue Degron Motif for Targeted Degradation — molecular glue degraders targeting faulty gene expression drivers in leukemia and lung cancer |
Equity Investment: Cognito Therapeutics — $105M Series C | Alzheimer's Disease | Pivotal Stage
Cognito Therapeutics | Equity Investment | Alzheimer's Disease | Pivotal (HOPE study)
| Parameter | Detail |
|---|---|
| Announced | March 5, 2026 |
| Round | Series C (oversubscribed) |
| Total raised | $105M |
| Cumulative raised | $233M |
| Lead investors | Morningside Ventures, IAG Capital Partners, Starbloom Capital |
| New investors | New Vintage, Apollo Health Ventures, Benvolio Group |
| HOPE pivotal study status | Fully enrolled (~673 patients, 70 sites) |
| Top-line data expected | Later in 2026 |
| Anticipated market entry | 2027 |
| FDA designation | Breakthrough Device |
Announced March 5, 2026, Cognito Therapeutics closed an oversubscribed $105M Series C to advance Spectris, its at-home neurostimulation platform for mild-to-moderate Alzheimer's disease. The round brings total capital raised to $233M and is positioned as a pre-commercialization financing bridging pivotal data readout to a planned 2027 U.S. market launch.
Spectris is an FDA Breakthrough Device-designated platform that delivers precisely timed gamma-frequency visual and auditory stimulation via a physician-prescribed at-home headset. The scientific basis draws from MIT research demonstrating that gamma oscillations — high-frequency brain wave patterns associated with memory and cognition — are disrupted in Alzheimer's disease, and that sensory stimulation at 40Hz can restore aspects of this neural network activity. Unlike anti-amyloid antibodies (lecanemab, donanemab), Spectris does not target a single protein pathway; it attempts to restore coordinated network function across multiple brain regions through native sensory pathways.
The pivotal HOPE study (NCT05637801) is fully enrolled with approximately 673 participants randomized to daily active stimulation or sham control for 12 months. Primary endpoints combine the ADCS-ADL (Activities of Daily Living) functional scale and MMSE cognitive scale using a combined statistical test. Top-line data are expected in the second half of 2026, with an FDA submission targeted shortly after.
The financing also supports expansion of the Spectris platform into additional neurodegenerative indications — including early exploration in Parkinson's disease — through a network of brain health collaboratories beginning with the WVU Rockefeller Neuroscience Institute.
The investment is strategically notable in the context of the current Alzheimer's landscape. Anti-amyloid antibodies have established proof of concept for disease modification but carry IV infusion requirements, ARIA monitoring obligations, and substantial administration burden. A home-based, non-pharmacologic, non-invasive device with a differentiated mechanism occupies a complementary niche that payers, health systems, and patients may find attractive if HOPE delivers positive data.
The oversubscription of this round, led by Morningside — a firm with deep CNS conviction — signals institutional confidence ahead of what will be a binary pivotal readout later this year.
Financing: Alithea Genomics — CHF 6.9M Seed Close | Biotech Tools / Transcriptomics | Early Stage | March 4, 2026
Alithea Genomics | Financing (Seed Round, Add-on Close) | Biotech Tools / RNA Transcriptomics for Drug Screening and Toxicology | Seed Stage | March 4, 2026
| Parameter | Detail |
|---|---|
| Announced | March 4, 2026 |
| Financing amount (add-on) | Additional CHF 3M |
| Total seed round | CHF 6.9M |
| Lead investor | Genku Ventures |
| Co-investors | Novalis Biotech, Zürcher Kantonalbank, and others |
| HQ | Lausanne, Switzerland (EPFL spin-out) |
Alithea Genomics, a Lausanne-based EPFL spin-out, announced an additional CHF 3M closing on its seed round, bringing the total to CHF 6.9M. The round was led by Genku Ventures, with co-investment from Novalis Biotech, Zürcher Kantonalbank, and additional undisclosed investors. The financing supports Alithea's BRB-seq platform, a bulk RNA sequencing technology designed for high-throughput drug screening and toxicology applications, enabling transcriptomic profiling at significantly lower cost and higher throughput than standard RNA-seq workflows.
The company's technology is positioned at the interface of genomics tools and pharmaceutical R&D services — the BRB-seq platform is used by pharmaceutical and biotech customers to assess drug effects on gene expression at scale, with applications in early discovery screening, mechanism-of-action profiling, and regulatory toxicology. The CHF-denominated round size and Swiss early-stage positioning mean this deal will not appear in most venture databases or deal aggregators that focus on USD-denominated or Series A+ financing events.
Clinical Data
Zealand Pharma petrelintide — Phase 2 ZUPREME-1 Top-Line Results
Zealand Pharma — petrelintide | Phase 2 Data | Obesity | Phase 2 | March 5–6, 2026
| Parameter | Detail |
|---|---|
| Study | ZUPREME-1 Phase 2 (NCT06662539; 493 participants, 33 sites) |
| Asset | Petrelintide (once-weekly amylin analogue) |
| Weight loss result | Up to 10.7% mean body weight reduction at 42 weeks (maximally effective dose arm) |
| Placebo result | 1.7% (p<0.001) |
| Tolerability | 4.8% discontinued due to AEs vs 4.9% placebo; no vomiting; no GI-related discontinuations at maximally effective dose |
| Market reaction | Zealand stock fell ~35% on data release |
| Investor expectation mismatch | Market had anticipated 13–20% weight loss comparable to GLP-1 competitors (semaglutide ~13–15%, tirzepatide ~20%) |
| Roche partnership value | Up to $5.3B: $1.65B upfront ($1.4B at closing + $250M in anniversary payments over two years), up to $1.2B development milestones, up to $2.4B sales-based milestones |
| Roche partnership CT-388 | Zealand pays Roche $350M for CT-388 contribution (offsetable against development milestones) |
| Profit sharing | 50/50 P&L sharing in U.S. and Europe; high single to low double-digit tiered royalties on rest-of-world net sales |
| Zealand advisors | Centerview Partners UK and Goldman Sachs International (financial); Goodwin Procter and Plesner Advokatpartnerselskab (legal) |
| Petrelintide patent | Composition-of-matter patent expires 2037 (with potential 5-year extension) |
| Next step | Phase 3 planned later in 2026; petrelintide/CT-388 combination Phase 2 planned H1 2026 |
ZUPREME-1 met all its endpoints, demonstrating meaningful and differentiated tolerability, but the 10.7% weight loss figure fell below the 13–20% range that investors and analysts had been modeling based on the GLP-1 class benchmark. Zealand's stock fell approximately 35% on the data release despite the clean safety profile. The Roche partnership — which values petrelintide at up to $5.3B in milestones and royalties across the combination and monotherapy development programs — remains intact. The clinical profile (lower efficacy but near-zero tolerability burden) positions petrelintide as a potential combination partner for GLP-1 agents rather than a standalone competitor.
AbbVie — SKYRIZI (risankizumab) AFFIRM Phase 3 Positive Topline Data | March 2, 2026
AbbVie — risankizumab (SKYRIZI) AFFIRM | Phase 3 Data | Moderate-to-Severe Crohn's Disease | Marketed Expansion | March 2, 2026
| Parameter | Detail |
|---|---|
| Announced | March 2, 2026 |
| Asset | Risankizumab (SKYRIZI) — subcutaneous induction formulation |
| Study | AFFIRM Phase 3 |
| Indication | Moderate-to-severe Crohn's disease |
| Co-primary endpoint 1 (Week 12) | CDAI clinical remission: 55% risankizumab vs. 30% placebo (p<0.0001) |
| Co-primary endpoint 2 (Week 12) | Endoscopic response: 44% risankizumab vs. 14% placebo (p<0.0001) |
| Maintenance outcomes (Week 24) | Clinical remission: 67% of responders; endoscopic response: 57% of responders |
| Population | Treatment-refractory: 50% had failed two or more prior advanced therapies |
| Commercial significance | SKYRIZI generated >$7B in global revenue in 2025 |
| Stock reaction | ABBV +~3.3% on announcement |
AbbVie reported positive topline data from the Phase 3 AFFIRM trial on March 2, 2026, demonstrating that a subcutaneous induction formulation of risankizumab met both co-primary endpoints in moderate-to-severe Crohn's disease. The study enrolled a heavily treatment-refractory population — half of participants had failed at least two prior advanced therapies — making the clinical response rates particularly meaningful in the context of limited remaining options for these patients.
The current SKYRIZI Crohn's disease approval uses intravenous induction at weeks 0, 4, and 8, with subcutaneous maintenance dosing. The AFFIRM data support a fully subcutaneous induction pathway, removing the IV infusion burden and potentially expanding patient access to community gastroenterology settings. For a drug generating over $7 billion annually — with Crohn's disease as one of its core growth indications alongside ulcerative colitis and plaque psoriasis — a label expansion enabling subcutaneous induction has material commercial implications for the AbbVie royalty and milestone stack.
Theravance Biopharma — 50% Workforce Reduction, R&D Shutdown
Theravance Biopharma | Corporate Restructuring | COPD | Commercial | March 3, 2026
Following a Phase 3 failure of its rare blood pressure disorder program, Theravance Biopharma announced a 50% workforce reduction and the complete shutdown of its R&D operations. The company will focus exclusively on commercial activity for its approved COPD treatment. The restructuring effectively converts Theravance from a development-stage company into a commercial-only specialty pharma business.
Roche — Fenebrutinib Phase III FENhance 1 — Relapsing Multiple Sclerosis | March 2, 2026
Roche reported positive Phase III data from the FENhance 1 study: fenebrutinib reduced relapses by 51% versus teriflunomide in relapsing multiple sclerosis. This is the third positive Phase III study for fenebrutinib, positioning it as the first BTK inhibitor with positive data in both relapsing and primary progressive MS.
Collaborations
Company Focus: Insilico Medicine
Insilico Medicine (HKEX: 03696) generated three distinct newsworthy developments during the week, spanning AI model development, clinical pipeline milestones, and regional market expansion.
Insilico Medicine / Liquid AI — LFM2-2.6B-MMAI Foundation Model | March 3, 2026
Insilico Medicine / Liquid AI — LFM2-2.6B-MMAI Foundation Model | Strategic Collaboration | AI for Drug Discovery | Cross-Stage
| Parameter | Detail |
|---|---|
| Partners | Insilico Medicine (HKEX: 03696) and Liquid AI (MIT-founded; private) |
| Deliverable | LFM2-2.6B-MMAI (v0.2.1) — 2.6B-parameter drug discovery foundation model; available now |
| Training data | ~120B pharmaceutical tokens across 200+ distinct tasks |
| Key benchmark results | Outperformed TxGemma-27B (10x larger) on 13 of 22 ADMET/PK tasks; up to 98.8% success rate on MuMO-Instruct multi-parameter molecular optimization; affinity prediction outperformed GPT-5.1, Claude Opus 4.5, and Grok-4.1 on Insilico's 2.5M-measurement internal benchmark |
| Deployment | On-premise / private pharmaceutical infrastructure (no external cloud required) |
| Financial terms | Not disclosed |
| Announced | March 3, 2026 |
Insilico Medicine and Liquid AI announced on March 3 a partnership that has produced LFM2-2.6B-MMAI, a single-checkpoint scientific foundation model trained to perform across the full drug discovery workflow — property prediction, ADMET profiling, molecular optimization, target-aware affinity scoring, functional group reasoning, and retrosynthesis planning — without requiring external cloud infrastructure. The model was trained on approximately 120 billion pharmaceutical tokens using Insilico's MMAI Gym, a training environment containing more than 1,000 pharmaceutical benchmarks.
The core innovation is architectural efficiency: a 2.6B-parameter Liquid Foundation Model (LFM) from MIT-founded Liquid AI, built on dynamical systems and signal processing rather than conventional transformer scaling, achieving performance competitive with models 10x larger. For pharmaceutical companies, this directly addresses the data-sovereignty constraint: firms with proprietary molecule, assay, and target data can now deploy frontier-class AI on private infrastructure rather than routing sensitive IP through public cloud APIs.
The partnership is strategically significant for Insilico in the context of its HKEX listing and its Pharmaceutical Superintelligence (PSI) roadmap. Demonstrating that Insilico's MMAI Gym can be used to train third-party model architectures — rather than only Insilico's own models — positions the platform as licensable infrastructure for AI labs and cloud providers, expanding the commercial thesis beyond Insilico's internal pipeline. The MMAI Gym is offered through flexible membership programs to pharma, biotech companies, AI labs, and cloud providers.
Insilico Medicine / TaiGen Biotechnology — ISM4808 Phase 1 First Patient Dosed | March 5, 2026
Insilico Medicine / TaiGen Biotechnology — ISM4808 | Milestone | Chronic Kidney Disease-Related Anemia | Phase 1 initiated
The TaiGen / Insilico Medicine licensing deal for ISM4808 — an AI-generated oral HIF-PHD inhibitor for CKD-related anemia, originally signed in December 2025 — hit its first contractual milestone on March 5, 2026, with TaiGen completing enrollment and dosing of the first Phase 1 subject. The underlying deal grants TaiGen exclusive rights to develop, commercialize, and sublicense ISM4808 across Greater China (mainland, Hong Kong, Macau, Taiwan), with Insilico eligible for upfront, development and sales-based milestones, and tiered royalties with a total disclosed value in the tens of millions of dollars. The Phase 1 study is a randomized, double-blind, placebo-controlled SAD/MAD design evaluating safety, tolerability, and pharmacokinetics in healthy adults.
TaiGen achieved first-patient dosing in under three months from license signing — a timeline Insilico's co-CEO Feng Ren described as a direct product of the AI-enabled discovery-to-clinic model. The deal is a relevant data point for the Insilico IPO narrative (HKEX: 03696) as the company builds its out-licensing revenue track record alongside its wholly owned pipeline.
Accord Healthcare / Foresee Pharmaceuticals — CAMCEVI 21mg | Regulatory Milestone | Advanced Prostate Cancer | Marketed (EU extension)
On March 6, 2026, the EMA's Committee for Medicinal Products for Human Use issued a positive opinion recommending approval of CAMCEVI 21mg — a 3-month ready-to-use subcutaneous leuprolide formulation — as an extension to the existing CAMCEVI marketing authorization for advanced prostate cancer. CAMCEVI is exclusively licensed to Accord Healthcare for European commercialization under a long-standing agreement with Foresee Pharmaceuticals (TPEx: 6576). The CHMP opinion advances to European Commission review, with a final marketing authorization decision expected in Q2 2026.
The 21mg 3-month formulation complements the existing 42mg 6-month version that launched in Europe in 2025, providing additional scheduling flexibility for prescribers and patients. No new financial terms are associated with this regulatory milestone; it triggers existing contractual milestone payments under the Accord/Foresee licensing agreement.
Insilico Medicine / Emirates Drug Establishment — AI Drug Discovery MoU | March 5–6, 2026
Insilico Medicine signed an MoU with the Emirates Drug Establishment to advance AI drug discovery tools in the UAE. Insilico shares rose 12.8% on the announcement. The company also plans to establish a Riyadh discovery cell in 2026, backed by Prosperity7 Ventures (Aramco).
| # | Partners | Type | Focus | Value | Date |
|---|---|---|---|---|---|
| 1 | J&J (Janssen) / Isomorphic Labs | AI drug discovery | Small molecules + biologics (multi-target) | Undisclosed | March 5 |
| 2 | Tempus AI / Merck | Data + AI biomarker | Oncology precision medicine; biomarker discovery | Undisclosed | March 3 |
| 3 | Hoth Therapeutics / OpenAI | AI workflow integration | KIT-driven cancers (GIST, systemic mastocytosis) | Undisclosed | March 4 |
| 4 | Various oncology collaborations | Research / licensing | Oncology targets (see section detail) | Various | W10 |
| 5 | Ono Pharmaceutical / Congruence Therapeutics | Drug discovery | Neurology + immunology | Undisclosed | W10 |
| 6 | Eli Lilly / WHO Foundation | Philanthropic | Obesity health systems strengthening | $1.68M | March 6 |
| 7 | CARsgen / Huadong Medicine + partners | Cell therapy portfolio | Multi-asset CAR-T portfolio | Undisclosed | W10 |
| 8 | Wolters Kluwer / Microsoft | Digital health integration | UpToDate x Dragon Copilot clinical workflow | Undisclosed | March 5 |
Royalty notes: J&J/Isomorphic — no royalty today; downstream licensing royalties contingent on programs advancing. Tempus/Merck — data access deal, no royalty structure. Lilly/WHO — philanthropic, no royalty. CARsgen partnerships — potential future royalty/milestone stacks as programs advance.
Notable Deals: AI Enters the Drug Development Workflow
| Parameter | Detail |
|---|---|
| Partners | Johnson & Johnson (Janssen R&D) and Isomorphic Labs (Alphabet/Google DeepMind spinout) |
| Announced | March 5, 2026 |
| Collaboration type | Cross-modality, multi-target drug discovery collaboration |
| Modalities | Small molecules AND biologics (broader than prior Isomorphic deals) |
| Financial terms | Undisclosed |
| Isomorphic prior deals | Eli Lilly (Jan 2024, up to $1.7B, small molecules only); Novartis (Jan 2024, up to $1.2B, small molecules only) |
| Differentiation | First Isomorphic collaboration explicitly covering biologics; multi-target scope exceeds prior deal structures |
| Isomorphic platform | AlphaFold-derived structure prediction and generative molecular design; integrates protein structure, binding affinity, and ADMET prediction |
| Strategic rationale for J&J | Janssen is building a computational drug discovery layer across its oncology, immunology, and neuroscience portfolios; Isomorphic partnership provides access to the most validated ML structure-prediction platform available |
Johnson & Johnson announced a drug discovery collaboration with Isomorphic Labs on March 5, 2026, marking the third major pharmaceutical partnership for the Alphabet/Google DeepMind spinout since its January 2024 deals with Eli Lilly and Novartis. The J&J deal is structurally broader than its predecessors: where Lilly and Novartis agreements covered small molecule discovery only, the Janssen partnership explicitly extends to biologics — monoclonal antibodies, bispecifics, and other protein-format therapeutics — reflecting Isomorphic's expanding capability set since AlphaFold3's release and its growing protein-protein interaction modeling capabilities.
Isomorphic Labs was spun out from Google DeepMind in 2021 specifically to apply AlphaFold protein structure prediction to drug discovery. Its platform integrates structure prediction, generative molecular design, and multi-parameter optimization in a computational workflow that can propose novel small molecule and biologic candidates for targets defined by the pharma partner. The J&J deal implies that Isomorphic can now predict how a biologic binder — an antibody or bispecific — would engage a protein target with sufficient accuracy to serve as a starting point for lead generation, a significant capability expansion beyond small molecule docking.
For royalty and deal tracking purposes, this collaboration is a revenue and milestone generator for Isomorphic (and by extension Alphabet), and a candidate for downstream licensing or royalty monetization should any programs advance to late-stage development.
Tempus AI / Merck — Expanded Multi-Year AI Biomarker Collaboration | March 3, 2026
| Parameter | Detail |
|---|---|
| Partners | Tempus AI, Inc. (Nasdaq: TEM) and Merck & Co., Inc. (NYSE: MRK) |
| Announced | March 3, 2026 |
| Type | Expanded multi-year research collaboration |
| Tempus offering | Access to de-identified multimodal patient data library + Lens Platform for AI-driven biomarker discovery |
| Merck focus | Oncology precision medicine; biomarker identification and patient stratification for clinical trials and commercial programs |
| Financial terms | Undisclosed |
| Tempus data assets | One of the largest de-identified real-world multimodal oncology databases in the U.S.: genomic, imaging, clinical, pathology, and outcomes data |
Tempus AI and Merck expanded their multi-year collaboration on March 3, 2026, with Merck gaining deeper access to Tempus's de-identified multimodal data library and AI-powered Lens Platform for oncology biomarker discovery. The collaboration follows Tempus's 2024 IPO and its positioning as an AI-driven precision medicine data company with one of the largest real-world oncology datasets in the U.S. Merck's use case centers on biomarker identification to stratify patients for clinical trials and support companion diagnostic development — a critical workflow as Merck defends and extends its Keytruda (pembrolizumab) franchise and builds next-generation oncology programs in the post-Keytruda patent cliff era. Financial terms were not disclosed, consistent with Tempus's practice of not breaking out individual deal economics ahead of its annual reporting.
Hoth Therapeutics / OpenAI | Collaboration | KIT-driven Cancers (Systemic Mastocytosis, GIST) | Preclinical
Announced March 4, 2026, this collaboration sits in an emerging category: a biopharmaceutical company deploying a large language model API directly into drug development operations rather than signing a conventional licensing or co-development agreement. Hoth has integrated OpenAI's API into the HT-KIT development workflow for preclinical data analysis, molecular modeling of KIT-driven pathways, and IND regulatory documentation preparation.
HT-KIT, an orphan-designated therapy targeting rare KIT-mutation-driven cancers including systemic mastocytosis and gastrointestinal stromal tumors, has shown >80% KIT mRNA/protein suppression in preclinical models with no dose-limiting toxicities reported. Statistically significant tumor volume reduction was observed by Day 8 in xenograft models. GLP-validated bioanalytical methods are complete ahead of IND submission.
Oncology Therapeutics: Collaborations and Licensing
CytoDyn / Creatv Bio | Collaboration | Metastatic Colorectal Cancer | Phase 2
CytoDyn is advancing leronlimab — a CCR5 antagonist with a complex regulatory history in HIV — into relapsed/refractory metastatic colorectal cancer through a collaboration with Creatv Bio. The scientific rationale centers on CCR5's role in tumor microenvironment immunosuppression and cancer cell migration. No financial terms disclosed.
Senhwa Biosciences / CellType | Collaboration | Oncology (Immuno-oncology, Tumor Microenvironment) | Phase 2/3
Senhwa's silmitasertib (CX-4945), a casein kinase 2 (CK2) inhibitor, is being combined with CellType's platform in an immuno-oncology collaboration targeting tumor microenvironment modulation. CK2 inhibition has been explored across multiple cancer types as a sensitizer for immune checkpoint therapy. No financial terms disclosed.
Asieris Pharmaceuticals / Photocure ASA | Regulatory Milestone / Active License | Cervical Intraepithelial Neoplasia (CIN2), HSIL | Approved (China, March 3, 2026)
See full entry under Asia-Pacific Regulatory and Market Events below.
Gene Solutions / ATC Genomics | Collaboration | Oncology
Vietnam-based genomics company Gene Solutions collaborates with ATC Genomics in an oncology genomics partnership. No financial terms disclosed.
Neurology, Imaging & Diagnostics
Siemens Healthineers / Cortechs.ai | Distribution | Neurological Conditions | Marketed
Siemens Healthineers adds Cortechs.ai's NeuroQuant Lesion Surveillance platform to its distribution network, covering Alzheimer's disease, ARIA (amyloid-related imaging abnormalities), multiple sclerosis, brain trauma, and other neurological conditions. NeuroQuant is an FDA-cleared automated volumetric MRI analysis tool. The timing is notable: ARIA monitoring has become a mandatory clinical workflow for Alzheimer's patients receiving anti-amyloid therapies (lecanemab, donanemab), creating a rapidly expanding market for automated lesion surveillance tools.
No financial terms disclosed.
LG Consulting Solutions / VentriPoint Diagnostics | Collaboration | Cardiac Imaging | Marketed
VentriPoint's VMS+ system uses artificial intelligence to construct 3D heart models from standard 2D echocardiography, enabling right ventricular volume assessment without MRI. The collaboration with LG Consulting Solutions is presumably focused on commercialization support. The technology addresses a real clinical gap: RV assessment via echo has historically been qualitative, and the ability to generate quantitative volumetric data without MRI is relevant in pediatric cardiology and pulmonary hypertension monitoring.
Therna Biosciences / Charles River | Collaboration | Ultra-Rare Lung Fibrosis, Lamb-Shaffer Syndrome | Preclinical / IND-enabling
Announced March 5, 2026, Therna Biosciences and Charles River are partnering to advance single-patient personalized RNA therapeutics — one mRNA program for an adult with rapidly progressive rare lung fibrosis, and one ASO program for a newborn with Lamb-Shaffer Syndrome, an ultra-rare CNS disorder. Therna's AI-enabled RNA design platform generated the lead mRNA candidate in three days, with laboratory validation in under three months. Charles River is conducting the preclinical development work, with the goal of filing a single-patient IND and dosing the lung fibrosis patient later in 2026.
The collaboration was unveiled at the Personalized Medicine World Conference on March 6. No financial terms disclosed.
XtalPi / ReviR Therapeutics | Collaboration | Charcot-Marie-Tooth Disease, Vanishing White Matter Disease | Phase 1
XtalPi, an AI-driven small molecule drug discovery platform company, collaborates with ReviR on RTX-117, targeting rare neurological diseases CMT and VWM. ReviR focuses on RNA-targeted therapeutics. The deal reflects continued computational/AI platform licensing into rare disease biotech development pipelines.
Collaboration: Ono Pharmaceutical / Congruence Therapeutics — Neurology and Immunology Drug Discovery
Ono Pharmaceutical / Congruence Therapeutics | Discovery Collaboration | Neurology, Immunology | Discovery | March 5, 2026
Ono Pharmaceutical and Congruence Therapeutics established a drug discovery collaboration focused on neurology and immunology target identification and candidate generation. Terms undisclosed.
Collaboration: Eli Lilly / WHO Foundation — Obesity Health Systems Strengthening | $1.68M Philanthropic Commitment | March 6, 2026
Eli Lilly and Company / WHO Foundation | Philanthropic Collaboration | Obesity / Metabolic Health | Global Health Systems | March 6, 2026
| Parameter | Detail |
|---|---|
| Announced | March 6, 2026 |
| Structure | Philanthropic collaboration; no commercial licensing or milestone terms |
| Lilly commitment | $1.68M through 2029 |
| Recipient | WHO Foundation (independent organization, Geneva; created 2020) |
| Focus | Assessing and strengthening health systems for obesity prevention and care in resource-limited settings |
| Lilly spokesperson | Patrik Jonsson, EVP and President, Lilly International |
| Financial/legal advisors | Not disclosed |
Announced March 6, 2026, Eli Lilly is committing $1.68M to the WHO Foundation through 2029 in support of the Foundation's efforts to strengthen health systems for obesity prevention and care. The collaboration is philanthropic rather than commercial — it carries no product licensing, milestone, or royalty terms — and is oriented toward building care pathways, assessing gaps, and supporting primary health care infrastructure in resource-limited settings where obesity prevalence is rising but treatment access is constrained.
The WHO Foundation is an independent organization headquartered in Geneva, established in 2020 to mobilize philanthropic capital in support of WHO's mission. It is legally and operationally distinct from the World Health Organization itself. The Lilly collaboration responds to the global obesity burden: more than 2.5 billion adults and over 390 million children and adolescents are overweight as of the most recent WHO data, with obesity prevalence having doubled in adults since 1990.
Longevity & Emerging Biology
Avaí Bio / Austrianova | Collaboration | Longevity, Anti-Aging, Alzheimer's, Cancer | Preclinical
Avaí Bio and Austrianova's SGAustria are collaborating on α-Klotho producing cells using Cell-in-a-Box encapsulated cell therapy technology. The program targets age-related disorders including Alzheimer's disease, cancer, and general longevity applications. Klotho is a protein with well-documented roles in aging and metabolic regulation; its therapeutic potential in neurodegeneration and systemic aging has attracted increasing research attention.
The preclinical stage and emerging biology put this in the watch-list category rather than near-term commercial relevance. No financial terms disclosed.
Collaboration: CARsgen Therapeutics / Huadong Medicine, Dispatch Bio, Shanghai Jingong, Zhuhai Hengqin SB Xinchuang — Multi-Asset Cell Therapy Portfolio
CARsgen Therapeutics Holdings Limited | Collaboration | Relapsed/Refractory Multiple Myeloma, Gastric/GEJ Adenocarcinoma, Hematologic Malignancies, Autoimmune Diseases, Solid Tumors | Multiple stages (Approved, Registration, Phase 1, Phase 2)
This entry reflects a cluster of collaboration agreements announced or confirmed in the context of CARsgen's 2025 Annual Results (March 6, 2026), spanning nine named assets and four distinct partners. The transactions are not a single omnibus agreement but rather a portfolio of partnerships that collectively define CARsgen's commercial and clinical development strategy across autologous, allogeneic, and in vivo CAR-T modalities. No aggregate financial terms have been disclosed across the partnership set.
Background: CARsgen Therapeutics (HKEX: 2171)
CARsgen Therapeutics is a Shanghai-headquartered biopharmaceutical company founded by Dr. Zonghai Li, a CAR-T pioneer and current CEO. The company has built an end-to-end CAR-T capability — target discovery, preclinical development, clinical execution, and commercial-scale manufacturing — with proprietary platforms spanning autologous, allogeneic, and in vivo modalities.
Its autologous programs are self-manufactured with proprietary plasmids and viral vectors, enabling what the company cites as a strong cost competitive advantage at the commercial stage. For 2025, CARsgen reported revenues of approximately RMB 125.7 million, gross profit of RMB 80 million, and a net loss of only RMB 103 million — an 87% reduction in losses year-over-year driven by zevor-cel commercialization.
Asset-by-Asset Breakdown
zevor-cel (zevorcabtagene autoleucel, CT053) | Autologous BCMA CAR-T | Approved (China), Phase 1b/2 (North America) | R/R Multiple Myeloma | Partner: Huadong Medicine
Zevor-cel is CARsgen's lead commercial product and the foundation of the Huadong Medicine partnership. It is a fully human, autologous CAR-T cell therapy targeting B-cell maturation antigen (BCMA), a protein highly expressed on malignant plasma cells in multiple myeloma and absent on most healthy tissues. The construct uses a fully human scFv (single-chain variable fragment) designated 25C2, which exhibits high binding affinity and a high monomer ratio — properties that contribute to potency and manufacturing consistency.
The mechanism follows the standard CAR-T workflow: a patient's T cells are collected via apheresis, genetically engineered ex vivo to express the BCMA-targeting chimeric antigen receptor, expanded, and reinfused as a single dose following lymphodepleting chemotherapy. BCMA expression on myeloma cells triggers receptor activation, inducing cytotoxic killing. The fully human antibody sequence is a deliberate design choice to reduce immunogenicity compared to murine-derived scFvs used in earlier-generation BCMA CAR-Ts such as ide-cel (BMS) and cilta-cel (J&J/Legend).
Zevor-cel was approved by China's NMPA in February 2024 for adult patients with R/R MM who have progressed after at least three prior lines of therapy (including a proteasome inhibitor and an immunomodulatory agent). Long-term follow-up from the Phase I portion of LUMMICAR-1, presented at IMS 2025 with 53.3 months median follow-up, showed no cases of grade 3 or higher CRS, no ICANS, and no delayed neurotoxicities — a safety profile that differentiates zevor-cel from some competing BCMA CAR-Ts. The Phase 1b/2 LUMMICAR STUDY 2 trial in North America is ongoing; CARsgen had planned a U.S.
BLA submission in the first half of 2025. Huadong Medicine received 218 confirmed orders in 2025 and holds exclusive commercialization rights for mainland China. Zevor-cel is also the CAR-T component in the Dispatch Bio DISP-11 combination program (see below).
satri-cel (satricabtagene autoleucel, CT041) | Autologous Claudin18.2 CAR-T | NDA under Priority Review (China), Phase 2 confirmatory | Gastric/GEJ Adenocarcinoma | Partner: Huadong Medicine
Satri-cel is a humanized autologous CAR-T product targeting Claudin18.2 (CLDN18.2), a tight junction protein that is a stomach-specific isoform of Claudin18. CLDN18.2 is highly expressed in gastric and gastroesophageal junction (GEJ) adenocarcinoma and, importantly, is largely absent from normal non-gastric tissue, giving it a favorable therapeutic window as a solid tumor target — one of the few solid tumor antigens amenable to CAR-T targeting without excessive on-target off-tumor toxicity.
Satri-cel is currently the world's first CAR-T cell therapy for solid tumors to advance to NDA review, a distinction that positions it as a landmark program for the field. China's CDE accepted the NDA for gastric/GEJ adenocarcinoma in June 2025, granted Breakthrough Therapy Designation in March 2025 and Priority Review in May 2025, with NMPA approval expected in the first half of 2026. The NDA was supported by data from the Phase Ib confirmatory ELIMYN18.2 (ST-02) trial in patients with advanced Claudin18.2-positive G/GEJA who had failed at least two prior lines of therapy.
Satri-cel is also being evaluated in pancreatic cancer, where CLDN18.2 expression is common. The FDA had previously placed a clinical hold on the U.S. IND, which was subsequently removed.
From a competitive standpoint, satri-cel enters a Claudin18.2-targeted landscape that also includes antibody-drug conjugates (ADCs) and bispecific antibodies — but remains the only CAR-T to reach NDA stage in this indication, and the first to demonstrate clinical validation of CLDN18.2 as a CAR-T target in humans.
CT0596 | Allogeneic BCMA CAR-T (THANK-u Plus™ platform) | Phase 1b/2 IND submitted (China), IIT ongoing | R/R Multiple Myeloma, Plasma Cell Leukemia (and future autoimmune applications) | Partner: Zhuhai Hengqin SB Xinchuang / UCARsgen JV
CT0596 is CARsgen's first allogeneic (off-the-shelf) BCMA CAR-T, developed on the proprietary THANK-u Plus™ platform. Unlike zevor-cel, which requires manufacturing from each individual patient's own T cells, CT0596 is derived from healthy donor T cells and engineered to be universally applicable — the central promise of allogeneic CAR-T as a way to reduce cost, turnaround time, and manufacturing complexity.
The THANK-u Plus™ platform achieves allogeneic compatibility through targeted gene knockouts: NKG2A, TRAC, and B2M genes are inactivated to reduce graft-versus-host disease (GvHD) risk and prevent host immune rejection of the donor-derived cells. First-in-human Phase 1 data from investigator-initiated trials were presented at ASH 2025 (December 2025) across 8 R/R MM patients who had received a median of 4.5 prior lines of therapy. The program demonstrated preliminary favorable tolerability and encouraging efficacy signals across all predefined dose levels, with measurable CAR-T expansion.
In separate earlier IIT data, two heavily pretreated plasma cell leukemia patients both achieved stringent complete responses. CARsgen submitted dual IND applications to the NMPA in December 2025 for Phase 1b/2 trials in R/R MM and primary plasma cell leukemia. Further investigation in autoimmune diseases mediated by autoreactive plasma cells is planned, mirroring the autoimmune expansion being pursued with CD19-directed allogeneic programs.
CT1190B | Allogeneic CD19/CD20 CAR-T (THANK-u Plus™ platform) | Phase 1 IIT (R/R NHL, SLE, SSc) | R/R Non-Hodgkin Lymphoma, Autoimmune Diseases | Partner: Zhuhai Hengqin SB Xinchuang / UCARsgen JV
CT1190B is an allogeneic dual-targeting CAR-T construct directed at both CD19 and CD20, covering the dominant B-cell surface antigens. The dual targeting strategy is intended to reduce antigen escape — a documented mechanism of relapse in single-target CD19 CAR-T therapies — by requiring tumor cells to simultaneously downregulate both targets to evade killing. Like CT0596, it is manufactured on the THANK-u Plus™ platform.
Clinical data presented in November 2025 from IITs in R/R NHL (14 patients enrolled as of October 17, 2025 across follicular lymphoma, mantle cell lymphoma, and DLBCL subtypes) showed that all three follicular lymphoma patients at the recommended lymphodepletion dose achieved complete responses, with 3 of 4 evaluable DLBCL patients achieving CR. Notably, no ICANS or graft-versus-host disease was observed — key safety signals for allogeneic products. CT1190B is also being evaluated in autoimmune disease, specifically moderate-to-severe refractory systemic lupus erythematosus (SLE) and refractory/progressive systemic sclerosis (SSc), in separate IITs.
The autoimmune application reflects the growing evidence that CD19 depletion — demonstrated most prominently by autologous products — can induce deep remissions in treatment-refractory lupus, and that an allogeneic version could dramatically expand patient access.
KJ-C2320 | Allogeneic CD38-targeting CAR-T | Phase 1 IIT initiated end-2024 | Acute Myeloid Leukemia | Partner: Zhuhai Hengqin SB Xinchuang / UCARsgen JV
CD38 is a glycoprotein widely expressed on AML blasts and, importantly, on plasma cells — making it a validated hematologic target (daratumumab, an anti-CD38 antibody, is a cornerstone of myeloma therapy). KJ-C2320 extends CAR-T targeting to CD38 in AML, an indication with high unmet need where CAR-T development has lagged behind lymphoma and myeloma programs. An investigator-initiated trial was initiated at end-2024.
KJ-C2526 | Allogeneic NKG2DL-targeting CAR-T | Preclinical/early IIT | AML, Other Malignancies, Senescence | Partner: Zhuhai Hengqin SB Xinchuang / UCARsgen JV
NKG2D ligands (NKG2DL) are stress-induced surface proteins upregulated on cancer cells and senescent cells. A CAR-T targeting NKG2DL is inherently tumor-agnostic in principle — it recognizes the stress signal rather than a lineage-specific antigen — making it potentially applicable across a broad range of malignancies and, notably, in clearance of senescent cells, a mechanism relevant to aging biology. This is an early-stage program but scientifically notable for its dual oncology and anti-senescence framing.
CT1390B | Allogeneic CLL1 (CLEC12A)-targeting CAR-T | Early clinical | Acute Myeloid Leukemia | Partner: Zhuhai Hengqin SB Xinchuang / UCARsgen JV
CLL1 (also called CLEC12A or CD371) is a myeloid surface antigen expressed on AML blasts and leukemic stem cells but not on normal hematopoietic stem cells, making it a target of considerable interest for AML-specific CAR-T. The selective expression on leukemic stem cells — the population responsible for disease relapse — is a particularly attractive property. CT1390B is an allogeneic CLL1-directed program on the THANK-u Plus™ platform.
KJ-C2527 | Allogeneic Claudin18.2-targeting CAR-T | Early-stage | Gastric Cancer | Partner: Zhuhai Hengqin SB Xinchuang / UCARsgen JV
KJ-C2527 is an allogeneic version of a Claudin18.2-directed CAR-T, targeting the same antigen as autologous satri-cel but manufactured from donor T cells for off-the-shelf deployment. If satri-cel demonstrates clinical success in gastric cancer, KJ-C2527 represents a next-generation, more accessible version of the same therapeutic concept, with the potential for broader reach at lower per-patient manufacturing cost.
KJ-C2529 | In Vivo CAR-T (CARvivo™ platform, CD19/CD20 target) | Preclinical | B-Cell Lymphoma | Partner: Zhuhai Hengqin SB Xinchuang / UCARsgen JV
KJ-C2529 represents CARsgen's most scientifically novel program: an in vivo CAR-T product candidate. Rather than manufacturing CAR-T cells outside the body and reinfusing them, in vivo CAR-T aims to deliver the CAR construct directly into the patient via a viral vector, reprogramming endogenous T cells in situ. CARsgen's proprietary platform for this approach is the lentiviral-based CARvivo™ system, which the company independently developed and describes as demonstrating excellent T cell transduction and targeting specificity.
KJ-C2529 targets CD19 and CD20 for B-cell lymphoma. In vivo CAR-T is widely viewed as the potential ultimate form factor for the modality — eliminating apheresis, ex vivo manufacturing, and the associated cost and delay — though the technical and regulatory challenges are substantial and no in vivo CAR-T has yet entered late-stage clinical development globally.
DISP-11 (Dispatch Bio Flare platform + zevor-cel) | Oncolytic Virus / CAR-T Combination | Phase 1 planned 2026 (China) | Solid Tumors of Epithelial Origin | Partner: Dispatch Bio
DISP-11 is a two-component investigational program pairing Dispatch Bio's Flare platform (specifically DV-10, the tumor-specific virus) with CARsgen's approved zevor-cel. The scientific problem it addresses is fundamental: standard CAR-T therapies have failed in solid tumors because solid tumors lack universally expressed tumor-specific surface antigens (unlike BCMA on myeloma cells) and maintain an immunosuppressive microenvironment that suppresses T cell function. The Flare platform is engineered to solve both problems simultaneously.
DV-10 is a tumor-selective oncolytic virus that systemically locates epithelial-derived tumor cells — which account for approximately 90% of all solid tumors — and delivers three payloads directly into them: a modified form of BCMA (called dBCMA), serving as a synthetic target antigen; the immune-stimulatory cytokine IL-18; and the chemokine CXCL9. The result is a two-step therapeutic sequence: DV-10 is administered first, "painting" tumor cells with the synthetic BCMA antigen while simultaneously activating and remodeling the tumor microenvironment; zevor-cel is then infused, with its BCMA-targeting CAR now able to recognize and kill the tagged tumor cells with specificity. The approach essentially converts a tumor without a natural CAR-T target into a tumor with an engineered target, while simultaneously creating a permissive immunological environment for T cell activity.
Dispatch Bio, founded in 2022 and based in Philadelphia and San Francisco, raised $216 million through a Series A with investors including ARCH Venture Partners, Parker Institute for Cancer Immunotherapy, Bristol Myers Squibb, and Alexandria Venture Investments. The company's parallel U.S. program, DISP-10, pairs DV-10 with BMS's approved ide-cel and received FDA IND clearance in January 2026. The China program, DISP-11, substitutes zevor-cel for ide-cel and is planned to initiate in 2026 through the CARsgen collaboration.
Preclinical data presented at SITC 2025 demonstrated potent anti-tumor responses across multiple epithelial tumor models with no activity in healthy cells.
Partnership Structure Context
The four partners in this collaboration set play distinct roles. Huadong Medicine (SZ: 000963) is CARsgen's established commercialization partner for mainland China, holding rights to zevor-cel and expected to hold rights to satri-cel upon approval. Shanghai Jingong Enterprise Development Co., Ltd. is a Jinshan District platform enterprise with which CARsgen signed strategic cooperation agreements to establish an advanced commercial manufacturing base for CAR-T products in Shanghai.
Zhuhai Hengqin SB Xinchuang Equity Investment Management Enterprise (a SoftBank Xinchuang-linked fund) entered agreements in February 2025 to co-invest in UCARsgen Biotech Limited — a joint venture entity dedicated to accelerating CARsgen's allogeneic CAR-T development in mainland China, which covers the THANK-u Plus™ and CARvivo™ pipeline assets (CT0596, CT1190B, CT1390B, KJ-C2320, KJ-C2526, KJ-C2527, KJ-C2529). Dispatch Bio is the U.S.-based combination therapy partner for the solid tumor Flare platform program. The aggregate structure represents a full-stack cell therapy ecosystem — commercialization, manufacturing infrastructure, development capital for next-generation modalities, and combination technology — assembled around CARsgen's proprietary platforms.
Partnership: Wolters Kluwer Health / Microsoft — UpToDate x Dragon Copilot Clinical Workflow Integration | Digital Health / Clinical Decision Support | March 5, 2026
Wolters Kluwer Health / Microsoft | Strategic Partnership / Product Integration | Clinical Decision Support, AI Workflow Automation | Marketed | March 5, 2026
| Parameter | Detail |
|---|---|
| Announced | March 5, 2026 |
| Deal value | Undisclosed |
| Structure | Product integration partnership; no equity or licensing terms disclosed |
| Parties | Wolters Kluwer Health (NL: WKL), Amsterdam; Microsoft Corporation (NASDAQ: MSFT), Redmond |
| Products | UpToDate (Wolters Kluwer) + Dragon Copilot (Microsoft) |
Wolters Kluwer Health and Microsoft announced an expansion of their existing partnership, integrating UpToDate — the market-leading clinical decision support platform used by clinicians in approximately 190 countries — with Microsoft's Dragon Copilot, the company's AI-powered clinical workflow automation assistant built on Azure AI and the Nuance Dragon ambient documentation platform. The integration embeds UpToDate clinical evidence directly into Dragon Copilot's workflow, enabling clinicians to access evidence-based guidance within ambient documentation and care coordination workflows rather than switching between separate systems.
No financial terms were disclosed. The partnership is a product integration rather than an equity or licensing transaction and will not appear in M&A deal databases by definition. Its inclusion here reflects the increasing relevance of AI workflow infrastructure deals to the broader healthcare technology landscape: UpToDate is embedded in clinical decision-making at scale, and its integration with ambient AI documentation tools represents a structural shift in how clinical evidence is accessed at the point of care.
Wolters Kluwer Health's broader digital transformation includes its ClinicalKey and Ovid platforms alongside UpToDate; the Microsoft partnership represents the most prominent AI integration announced for the UpToDate product to date.
Supply Agreements
Supply Agreement: Fujifilm Vet Systems / VolitionRx — Nu.Q® Vet Cancer Test | Marketed
Fujifilm Vet Systems Co. Ltd / VolitionRx Limited | Supply Agreement | Cancer Diagnostics in Dogs | Marketed
Fujifilm Vet Systems Co. Ltd has entered into a supply agreement with VolitionRx for the Nu.Q® Vet Cancer Test, a blood-based assay for cancer detection in dogs. Nu.Q® measures nucleosome levels as cancer biomarkers, providing veterinarians with a non-invasive screening tool for canine oncology.
The agreement expands the commercial distribution footprint for Nu.Q® Vet through Fujifilm's veterinary diagnostics network. No financial terms disclosed. The companion animal diagnostics market has attracted increasing deal activity as precision medicine concepts migrate from human medicine into veterinary applications.
Supply Agreement: Boops Pets / Ixoreal Biomed — KSM-66 Ashwagandha (Stress and Anxiety in Dogs) | Marketed
Boops Pets / Ixoreal Biomed | Supply Agreement | Stress and Anxiety (Companion Animals) | Marketed
Boops Pets has entered into a supply agreement with Ixoreal Biomed for KSM-66 Ashwagandha, a branded, full-spectrum ashwagandha root extract, for use in stress and anxiety management products for dogs. KSM-66 is a clinically studied ingredient with human-medicine validation for cortisol modulation and stress reduction; its application in companion animal health reflects the broader trend of evidence-based nutraceutical ingredients crossing over from human wellness into the pet supplement market. No financial terms disclosed.
The companion animal functional wellness segment is one of the fastest-growing categories in pet health, driven by owner humanization trends and demand for non-pharmaceutical anxiety management solutions.
FDA Regulatory Actions
| # | Company | Product | Indication | Action | Royalty / Deal Relevance | Date |
|---|---|---|---|---|---|---|
| 1 | J&J (Janssen) | Tec-Dara (teclistamab + daratumumab) | R/R multiple myeloma (≥1 prior line) | NDA approval (3rd National Priority Voucher) | No royalty monetization disclosed; J&J owns both components | March 5 |
| 2 | Bristol Myers Squibb | Sotyktu (deucravacitinib) | Psoriatic arthritis (sNDA) | sNDA approval — first TYK2 inhibitor in PsA | No royalty disclosed; BMS full owner | March 6 |
| 3 | Lantheus | PYLARIFY TruVu (piflufolastat F 18) | Prostate cancer PSMA PET imaging | NDA approval (505(b)(2)) — optimized formulation | No royalty monetization disclosed | March 6 |
| 4 | Novo Nordisk | Sogroya (somapacitan-beco) | Three pediatric indications (GH deficiency) | sNDA approval | No royalty disclosed | W10 |
| 5 | BioMarin | Palynziq (pegvaliase-pqpz) | Pediatric PKU (ages 12+) | Label expansion | No royalty disclosed | W10 |
| 6 | Hemab Therapeutics | Sutacimig | Glanzmann thrombasthenia | Breakthrough Therapy Designation | Partnering/licensing candidate | W10 |
| 7 | Cara Medical | CARA System | Cardiac conduction visualization | 510(k) clearance | MedTech | W10 |
| 8 | Bayer | MEDRAD MRXperion | 7T MRI compatibility | 510(k) clearance | MedTech | W10 |
Royalty notes: Tec-Dara is fully J&J-owned. Sotyktu is fully BMS-owned — no external royalty stream known. PYLARIFY TruVu: Lantheus developed piflufolastat F 18 internally; no disclosed external royalty obligation. Sogroya/BioMarin Palynziq: no disclosed royalty streams. Hemab sutacimig: privately held, partnering candidate — BTD increases asset attractiveness for licensing/royalty deal.
FDA Approval: Johnson & Johnson Tec-Dara (teclistamab + daratumumab) — Relapsed/Refractory Multiple Myeloma
Johnson & Johnson — Tec-Dara | FDA Approval | Relapsed/Refractory Multiple Myeloma | Marketed | March 5, 2026
| Parameter | Detail |
|---|---|
| Approved | March 5, 2026 |
| Asset | Tec-Dara (teclistamab + daratumumab hyaluronidase-fihj) |
| Indication | Relapsed/refractory multiple myeloma in adults who received ≥1 prior line of therapy |
| Key efficacy data | 83% reduction in risk of disease progression or death versus standard of care (Phase 3) |
| Review pathway | FDA National Priority Voucher (CNPV) program; decision rendered 55 days after filing |
| Regulatory note | Third approval under the CNPV program |
Tec-Dara — a combination of J&J's BCMA-targeting bispecific antibody teclistamab with daratumumab hyaluronidase — received FDA approval on March 5, 2026 for adults with relapsed/refractory multiple myeloma who have received at least one prior line of therapy. Phase 3 data demonstrated an 83% reduction in risk of disease progression or death versus standard of care. The approval was the third granted under the FDA's National Priority Voucher program, with a decision rendered just 55 days after filing. This approval expands the multiple myeloma treatment landscape across a patient population also addressed by recently approved BCMA-directed CAR-T therapies.
FDA Approval: Bristol Myers Squibb Sotyktu (deucravacitinib) sNDA — Psoriatic Arthritis
Bristol Myers Squibb — Sotyktu | FDA sNDA Approval | Psoriatic Arthritis | Marketed Expansion | March 6, 2026
| Parameter | Detail |
|---|---|
| Approved | March 6, 2026 (PDUFA action date) |
| Asset | Sotyktu (deucravacitinib) |
| New indication | Active psoriatic arthritis in adults |
| Designation | First TYK2 inhibitor approved for psoriatic arthritis |
| Phase 3 data (PsA-1) | ACR20 response: 54.2% vs 34.1% placebo at Week 16 |
| Basis | POETYK PsA-1 and PsA-2 Phase 3 trials |
The FDA approved the Sotyktu sNDA on its PDUFA action date of March 6, 2026, making deucravacitinib the first TYK2 inhibitor approved for psoriatic arthritis. The approval is based on results from the POETYK PsA-1 and PsA-2 Phase 3 trials, which showed ACR20 response rates of 54.2% versus 34.1% placebo at Week 16 in PsA-1. Sotyktu was previously approved for plaque psoriasis; the PsA expansion broadens the addressable population and strengthens BMS's immunology franchise against established IL-17 and IL-23 inhibitors in the PsA market.
FDA Approval: Lantheus PYLARIFY TruVu (piflufolastat F 18) — PSMA PET Imaging
Lantheus — PYLARIFY TruVu | FDA NDA Approval (505(b)(2)) | Prostate Cancer PSMA PET Imaging | Marketed Expansion | March 6, 2026
| Parameter | Detail |
|---|---|
| Approved | March 6, 2026 |
| Asset | PYLARIFY TruVu (piflufolastat F 18) |
| Indication | PSMA PET imaging in men with prostate cancer |
| Pathway | 505(b)(2) |
| Formulation improvement | ~50% larger batch size; improved radioactive concentration |
| Clinical data | 96% specificity, 85–87% correct localization rate |
| Launch timing | Planned Q4 2026 |
Lantheus received FDA approval for PYLARIFY TruVu via the 505(b)(2) pathway on March 6, 2026. The new formulation increases batch size by approximately 50% and improves radioactive concentration relative to the original PYLARIFY, with 96% specificity and 85–87% correct localization rate. Commercial launch is planned for Q4 2026. The improvement addresses manufacturing scalability constraints in the original formulation and positions Lantheus to capture growing clinical demand for PSMA PET imaging as prostate cancer treatment volumes increase with expanded access to lutetium-177 PSMA therapy (Pluvicto).
FDA sNDA Approval: Novo Nordisk Sogroya (somapacitan-beco) — Three Pediatric Indications
Novo Nordisk — Sogroya | FDA sNDA Approval | Idiopathic Short Stature, SGA, Noonan Syndrome | Approved Feb 27 / Reported March 2, 2026
| Parameter | Detail |
|---|---|
| Approved | February 27, 2026 |
| Asset | Sogroya (somapacitan-beco) |
| New indications (3) | Idiopathic short stature; short stature born small for gestational age; growth failure associated with Noonan syndrome |
| Population | Children ≥2.5 years |
| Clinical basis | Phase 3 REAL8 basket study (n=307) |
| Designation | First long-acting once-weekly growth hormone approved for all three conditions |
Note: Approved February 27, 2026; included given widespread reporting during the March 2–7 window and relevance to Novo Nordisk's endocrinology franchise.
Novo Nordisk received FDA approval for three new pediatric indications for Sogroya: idiopathic short stature, short stature born small for gestational age, and growth failure associated with Noonan syndrome — all for children aged 2.5 years and older. Based on the Phase 3 REAL8 basket study (n=307), Sogroya becomes the first once-weekly growth hormone approved across all three conditions, consolidating its position against daily recombinant hGH products across multiple pediatric growth disorder indications.
FDA Label Expansion: BioMarin Palynziq (pegvaliase-pqpz) — Pediatric PKU (Ages 12+)
BioMarin — Palynziq | FDA sNDA Label Expansion | Phenylketonuria | Marketed Expansion | ~March 2, 2026
BioMarin's Palynziq (pegvaliase-pqpz) received an FDA label expansion to include patients aged 12 and older with phenylketonuria (PKU). The product was previously approved only for adult patients. The expansion extends commercial reach into the adolescent PKU population, an important access gap given the chronic nature of PKU management and the early onset of the disease.
FDA Breakthrough Therapy Designation: Hemab Therapeutics sutacimig — Glanzmann Thrombasthenia
Hemab Therapeutics — sutacimig | FDA Breakthrough Therapy Designation | Glanzmann Thrombasthenia | Phase 1/2 | March 5, 2026
The FDA granted Breakthrough Therapy Designation to Hemab Therapeutics' sutacimig for the prevention of bleeding episodes in Glanzmann thrombasthenia, an ultra-rare inherited platelet disorder. Sutacimig is a first-in-class bispecific antibody targeting FVIIa and TLT-1, designed to restore hemostasis independent of platelet function. The designation was based on Phase 1/2 data and adds to Hemab's existing Fast Track and Orphan Drug designations for the program.
FDA 510(k) Clearance: Cara Medical CARA System — Cardiac Conduction Visualization
Cara Medical — CARA System | FDA 510(k) Clearance | Cardiac Conduction / Electrophysiology | Device | March 2, 2026
Cara Medical received FDA 510(k) clearance for the CARA System, a CTA-based platform for noninvasive 3D visualization of the cardiac conduction system. The device had previously received FDA Breakthrough Device designation. The CARA System enables pre-procedural cardiac mapping without invasive electrophysiology study, with potential applications in arrhythmia ablation planning and structural heart procedures.
FDA 510(k) Clearance: Bayer MEDRAD MRXperion — 7T MRI Compatibility
Bayer / MEDRAD — MRXperion | FDA 510(k) Clearance | MR Imaging / Contrast Injection | Device Expansion | March 4, 2026
Bayer's MEDRAD MRXperion MR contrast injection system received FDA 510(k) clearance for expanded field strength compatibility up to 7 Tesla and a new ISI2 scanner interface. The 7T expansion is relevant given growing clinical deployment of ultra-high-field MRI systems, which offer enhanced spatial resolution for neuroimaging, cardiovascular, and oncology applications. The ISI2 interface improves scanner-injector communication across installed base systems.
FDA Enforcement and Regulatory Disputes
UniQure / FDA — AMT-130 Huntington's Gene Therapy Dispute | March 2–6, 2026
UniQure disclosed that FDA rejected its plan to file a BLA for AMT-130 based on Phase I/II data with external controls, instead issuing a strong recommendation for a prospective, randomized, sham surgery-controlled Phase 3. An FDA official stated publicly that the agency is not convinced therapeutic benefit has been established. UniQure shares fell approximately 40% on the disclosure. The company plans a Type B meeting in Q2 2026 to discuss Phase 3 design. UniQure separately faces a clinical hold on its Fabry disease program. The combined news materially extends the financing runway requirements for a company that had positioned AMT-130 as a near-term regulatory catalyst.
FDA — 30 Warning Letters to Telehealth Compounders | March 3, 2026
FDA issued 30 warning letters to telehealth companies over false and misleading claims about compounded GLP-1 products. Commissioner Makary characterized the action as a new era of enforcement, noting the agency had issued more letters in six months than in the entire prior decade. GenoGenix was specifically flagged for repackaging semaglutide, tirzepatide, and retatrutide (still in development), with three documented patient ER visits. FDA also issued an untitled letter to Novo Nordisk regarding misleading superiority claims in its "Only One Ozempic" campaign. The enforcement wave is directly relevant to the royalty market: compounders had been suppressing branded GLP-1 demand, and the enforcement acceleration supports the royalty cash flow trajectory of royalty holders on Ozempic, Wegovy, and Mounjaro.
FDA — Request for Information on In-Home Opioid Disposal | March 5, 2026
FDA issued an RFI seeking comment on potential new standards requiring sponsors to make in-home opioid disposal systems available through dispensers. Comments due April 6, 2026.
EveryOne Medicines — Shutdown | March 3, 2026
EveryOne Medicines shut down one week after FDA published its Plausible Mechanism Framework for individualized therapies. The bespoke gene therapy startup — founded by Julia Vitarello following her daughter's death from Batten disease — found the framework's requirement that each individualized medicine be submitted as a discrete application commercially unsustainable. HHS disputed any link between the framework and the shutdown.
Alltrna — 34% Workforce Reduction | March 5, 2026
Flagship Pioneering-backed tRNA therapeutics company Alltrna reduced its workforce by approximately 34% (19 of ~55 employees), its second reduction in under a year. The company stated the cuts would accelerate its path to the clinic with its first engineered tRNA drug candidate.
BioAtla — ~70% Workforce Reduction + Strategic Review | March 2, 2026
BioAtla, an antibody therapeutics company focused on conditionally active biologic (CAB) technology, announced approximately 70% workforce reduction and a formal strategic review process that includes exploring asset sales, licensing, and partnership options across its nine preclinical and clinical-stage solid tumor programs. This follows a prior 30% reduction in March 2025 and represents a near-total dismantling of the company's operating structure. BioAtla's CAB platform is based on antibodies that become active only in the tumor microenvironment's low-pH conditions, a differentiated mechanism with licensing potential if validated programs are transferred to well-capitalized acquirers.
Kyowa Kirin / Amgen — Rocatinlimab (OX40 mAb) Full Program Discontinuation | March 3, 2026
| Parameter | Detail |
|---|---|
| Asset | Rocatinlimab (anti-OX40 monoclonal antibody; enhanced ADCC) |
| Developer | Kyowa Kirin Co., Ltd. (TSE: 4151); former development partner Amgen |
| Decision date | March 3, 2026 |
| Decision | All ongoing clinical trials discontinued; full program halted |
| Safety signal | 1 new confirmed + 1 suspected Kaposi's sarcoma case, in addition to 1 previously confirmed case — three cases total across the program |
| Mechanism concern | OX40 pathway modulation may impair antiviral immune surveillance (HHV-8/Kaposi's sarcoma biologically plausible link) |
| Overall malignancy rate | Below expected background rates — but case characteristics raised a plausible biological concern that could not be excluded |
| Indications being studied | Moderate-to-severe atopic dermatitis (Phase 3, primary indication); prurigo nodularis (Phase 2/3); moderate-to-severe asthma (Phase 2/3) |
| Phase 3 program | ROCKET-IGNITE and ROCKET-HORIZON in ~1,500 adults; both trials had met primary endpoints (rIGA response) |
| Regulatory submission timeline | Had been planned for FDA submission H1 2026 — now abandoned |
| Amgen background | Amgen had already exited its commercialization partnership (~$400M original deal) approximately one month earlier in late January 2026 |
| Kyowa Kirin stance | "Likelihood of continuing development or resuming clinical trials to be extremely low" |
| Next steps | Investigators and regulators being notified; all studies terminated after safety follow-up completion; full program dataset analysis planned |
Kyowa Kirin announced on March 3, 2026 the discontinuation of all clinical trials for rocatinlimab, eliminating the most advanced OX40-targeted therapy in inflammatory disease development. The decision followed a joint safety committee meeting between Kyowa Kirin and Amgen in late February 2026, which identified a third Kaposi's sarcoma case — one new confirmed and one suspected, in addition to a previously confirmed case — across the global rocatinlimab clinical program.
The OX40 pathway is a costimulatory receptor on activated T cells that controls T cell activation, survival, and memory formation. Rocatinlimab targets OX40 with enhanced antibody-dependent cellular cytotoxicity (ADCC) to deplete pathogenic memory T cells implicated in atopic dermatitis. The same depletion mechanism creates an immunological vulnerability: OX40-mediated pathways contribute to antiviral immune surveillance, and Kaposi's sarcoma — caused by human herpesvirus-8 (HHV-8), a virus kept latent by intact T cell immunity — is a biologically plausible consequence of disrupting that surveillance. Although the overall malignancy count remains below statistical background rates, the mechanistic coherence of the Kaposi's sarcoma signal — combined with three cases across what was already a large Phase 3 program — was sufficient for both companies to conclude that risks may outweigh benefits.
The discontinuation is consequential beyond Kyowa Kirin's balance sheet. It eliminates the only late-stage OX40 inhibitor in development for inflammatory diseases and raises a class-level safety question for the target. Sanofi's amlitelimab, an anti-OX40L antibody (blocking the ligand rather than the receptor), recorded one Kaposi's sarcoma case in its development program, prompting Jefferies analysts to downgrade commercial expectations to a niche population. The OX40/OX40L axis — once viewed as a promising "upstream" mechanism for durable T cell reset in chronic inflammatory disease, potentially superior to downstream cytokine blockers — is now effectively derisked as a development priority for the foreseeable future.
From a deal intelligence perspective, the termination removes a ~$400M Amgen partnership asset from the landscape. The original Amgen-Kyowa Kirin collaboration (2021, approximately $400M upfront + milestones) had already been unwound by Amgen in January 2026 — a leading indicator that Amgen had already seen the safety data trajectory. Royalty investors and biopharma deal teams evaluating OX40-mechanism assets should treat the Kaposi's sarcoma signal as a class-level risk factor pending the full program dataset analysis.
Science Corp — $230M Series C | Neurotech / BCI | March 5, 2026
Science Corp (privately held), the brain-computer interface and neurotech company co-founded by Max Hodak, closed a $230 million Series C financing round — one of the week's largest single venture financings across all of life sciences. Science Corp is developing the Science Eye visual prosthesis (a high-resolution retinal implant for blindness) and a suite of high-bandwidth neural interface devices. The round positions the company at the neurotech boundary of the pharmaceutical and medical device sectors, with potential royalty and licensing implications as platform-based neural recording technologies enter clinical validation.
Atavistik Bio — $40M Series B Extension | March 5, 2026
RA Capital led a $40 million extension to Atavistik Bio's Series B, bringing the total to $160 million. Funds support clinical development of ATV-1601, an oral AKT1-selective inhibitor in development for hereditary hemorrhagic telangiectasia, alongside a JAK2 V617F mutant-selective inhibitor program for myeloproliferative neoplasms. The rare disease focus and precision kinase selectivity position Atavistik's assets as potential out-licensing candidates.
Bavarian Nordic — CEO Paul Chaplin Stepping Down | Early March 2026
Paul Chaplin announced plans to step down as CEO of Bavarian Nordic by end of 2026, citing personal reasons (family relocation to Australia) after 12 years leading the company through the development and commercialization of its Jynneos mpox and smallpox vaccine franchise. The board has initiated a CEO search. Chaplin's tenure covered Jynneos's FDA approval, emergency use authorization for the mpox outbreak, and substantial revenue growth. His departure triggers a leadership transition at a company with significant royalty and milestone obligations tied to its U.S. government vaccine contracts.
Pierre Fabre — Type A Meeting Request for Tabelecleucel | March 2–3, 2026
Pierre Fabre submitted a Type A meeting request to FDA regarding tabelecleucel (Ebvallo), its allogeneic T-cell therapy for EBV-positive PTLD, following a second Complete Response Letter in January 2026. Ebvallo is already approved in the EU, UK, and Switzerland.
BridgeBio — BBP-418 (ribitol) Phase 3 FORTIFY — Late-Breaking Data Confirms Efficacy | LGMD2I/R9 | March 4, 2026
BridgeBio announced that its FORTIFY Phase 3 trial evaluating BBP-418 (ribitol) in limb-girdle muscular dystrophy type 2I/R9 (LGMD2I/R9) met its pre-specified efficacy endpoints, with additional interim analysis data accepted as a late-breaking oral presentation at the MDA Clinical & Scientific Conference (March 8–11, 2026). An NDA filing is targeted for H1 2026; if approved, BBP-418 would become the first approved therapy for LGMD2I/R9, a rare inherited muscle-wasting disease caused by FKRP gene mutations affecting approximately 8,000–10,000 U.S. patients. The program holds Breakthrough Therapy Designation. BridgeBio retains full commercial rights with no disclosed external royalty obligations.
DBV Technologies — VIASKIN Peanut Phase 3 VITESSE Additional Data | AAAAI 2026 | March 1–2, 2026
Additional analyses from DBV Technologies' VITESSE Phase 3 trial of VIASKIN Peanut Patch, presented at AAAAI 2026, showed approximately 83% of treated children increased their eliciting dose at Month 12 versus approximately 48% in the placebo group, while only 6.4% of treated patients experienced a decreased eliciting dose compared to 24% placebo. The patch was well tolerated with primarily mild local reactions at the application site. VIASKIN Peanut is a transcutaneous immunotherapy under FDA regulatory review for peanut allergy in children, with a distinctive delivery modality that carries potential licensing and royalty implications.
Quell Therapeutics (UK) — Dual Clinical Milestone: LIBERATE Data + CHILL Trial Initiation | March 3, 2026
UK-based Quell Therapeutics achieved two simultaneous clinical milestones on March 3. First, the company released LIBERATE Phase 1/2 interim data for QEL-001, its first-in-class anti-HLA-A2 CAR-Treg cell therapy being evaluated in liver transplant recipients to induce immune tolerance: 9 patients enrolled, no serious adverse events, and evidence of durable CAR-Treg persistence in peripheral blood. Second, Quell simultaneously initiated the CHILL Phase 1/2 trial of QEL-005, a CD19-targeting CAR-Treg, in patients with refractory rheumatoid arthritis and systemic sclerosis, across sites in the UK, Germany, and Spain. The combination of first-in-human tolerability data and initiation of a second program in a separate indication on the same day represents a significant milestone cluster for a pre-commercial UK cell therapy company with backing from leading life sciences investors.
Theolytics (UK) — €8M Horizon Europe Grant | OCTOPOD-IV Trial | March 4, 2026
Oxford-based oncolytic immunotherapy company Theolytics was awarded an €8 million grant from Horizon Europe to fund a Phase 2a expansion of its OCTOPOD-IV trial evaluating THEO-260, its lead intravenous oncolytic adenovirus, in platinum-resistant ovarian cancer. Horizon Europe grants are non-dilutive public funding; this award extends Theolytics' clinical runway and validates the program for potential partnership discussions. Platinum-resistant ovarian cancer remains an indication with limited effective options and significant commercial opportunity for mechanisms demonstrating durable responses.
HUTCHMED — HMPL-A580 Phase I/IIa First Patient Dosed | March 4, 2026
HUTCHMED (AIM: HCM), the UK/Hong Kong-listed oncology company with operations in China, dosed the first patient in the HMPL-A580 Phase I/IIa first-in-human trial in patients with advanced solid tumors. HMPL-A580 uses HUTCHMED's proprietary Antibody-Targeted Therapy Conjugate (ATTC) platform — a distinctive modality that pairs targeted therapy agents as the payload rather than traditional cytotoxic chemotherapy, representing a next-generation ADC architecture. The ATTC approach targets the tumor with therapeutic precision at multiple levels simultaneously. No financial terms disclosed.
Alvotech — CEO Transition | March 2026 (Effective End of March)
Iceland-headquartered biosimilars specialist Alvotech named Lisa Graver as its next CEO, with founder Robert Wessman transitioning to Executive Chairman. Graver previously led Alvogen until its acquisition by Lotus Pharmaceutical in December 2025. The transition represents a planned handover of operational leadership as Alvotech continues to expand its biosimilar portfolio in the U.S. and European markets.
GSK — Workforce Reduction (~350 positions) | Early March 2026
GSK reduced approximately 350 positions across U.S. and U.K. operations as part of portfolio and investment reprioritization. Fewer than 50 U.K. roles were affected. The reduction is consistent with GSK's stated strategy of focusing resources on priority therapeutic areas and reflects broader industry-wide pressure on headcount in commercial and non-priority research functions.
EMA and International Regulatory
| # | Company | Product | Active Substance | Opinion / Action | Royalty / Deal Relevance |
|---|---|---|---|---|---|
| 1 | Moderna | mCOMBRIAX | mRNA combo (COVID-19 + influenza) | Positive — world's first combined COVID/flu mRNA vaccine; adults ≥50 | Settlement with Arbutus/Genevant (W10 feature) extinguishes all future LNP royalties on mCOMBRIAX; Arbutus/Genevant receive lump sum only — no ongoing royalty stream from EU sales |
| 2 | Ipsen / Day One | Ojemda | Tovorafenib | Positive (conditional MA) — pediatric LGG ≥6 months | Servier acquires inbound double-digit tiered royalty stream from Ipsen on ex-U.S. net sales via Day One acquisition; exact rate disclosed only as "double-digit tiered" |
| 3 | Crinetics | Palsonify | Paltusotine | Positive — acromegaly (oral SRL) | No disclosed royalty obligation |
| 4 | Novartis | Rhapsido | Remibrutinib | Positive — chronic spontaneous urticaria (BTK inhibitor) | No disclosed royalty; Novartis developed internally |
| 5 | X4 Pharmaceuticals | Xolremdi | Mavorixafor | Exceptional circumstances — WHIM syndrome (ages ≥12) | No disclosed royalty; X4 owns asset |
| 6 | Tanabe Pharma | Onerji | Levodopa/carbidopa | Positive — advanced Parkinson's (motor fluctuations) | No disclosed royalty |
| 7 | Sanofi / DNDi | Acoziborole | Acoziborole | EU-M4All approval — sleeping sickness (single-dose oral) | Public health / access pricing; not commercially royalty-relevant |
| 8 | Sanofi | Dupixent extension | Dupilumab | Positive — pediatric CSU ages 2–11 (first targeted EU medicine for this age group) | Regeneron earns a 35–45% sliding-scale share of ex-U.S. Dupixent operating profit (not a royalty on net sales); FDA decision on same pediatric CSU population expected April 2026 |
| 9 | Acadia | Daybu | Trofinetide | Negative opinion | No royalty implication; EU market closed |
| 10 | Vanda | iloperidone | Iloperidone | Negative opinion (3rd rejection) | No royalty implication |
| 11 | Pfizer | Zumrad | Sasanlimab | Withdrawal — bladder cancer | No royalty implication |
Royalty notes: The Ojemda EMA positive opinion is directly consequential for the Servier/Day One deal: Ipsen owes double-digit tiered royalties to Day One (now Servier) on all ex-U.S. Ojemda net sales; the CHMP positive opinion confirms that EU commercial revenue — and therefore the inbound royalty stream — is coming live, and the stream transfers to Servier intact with no third-party fund holding an interest. The publicly disclosed rate is "double-digit tiered"; the "mid-teens" characterization cited in analyst commentary has not been confirmed in any public filing and should be treated as an estimate.
Dupixent EU pediatric CSU label extension: Regeneron's economic interest in ex-U.S. Dupixent sales is a profit share under the 2007 collaboration agreement, not a royalty on net sales. Sanofi records global Dupixent net product sales; Regeneron receives 35% of ex-U.S. operating profit, scaling to 45% at higher sales volumes (Sanofi retains 65–55%). The pediatric CSU label expansion for ages 2–11 modestly increases the EU royalty-eligible population. A parallel FDA sBLA for the same pediatric CSU population is under review with a decision expected April 2026, which would expand the 50/50 U.S. profit-share base. Note: Regeneron filed suit against Sanofi in November 2024 alleging Sanofi breached the collaboration agreement by denying access to PBM pricing contracts; Regeneron's independent audit already identified a monetary adjustment owed to it, and the litigation remains active — the collaboration profit-split economics are disputed.
mCOMBRIAX EMA positive opinion: the W10 Moderna/Arbutus-Genevant settlement extinguishes all future LNP royalties on Moderna's infectious disease vaccine pipeline, including mCOMBRIAX, in exchange for a $950M lump-sum payment (plus up to $1.3B contingent on a Federal Circuit appeal). The settlement grants Moderna a global royalty-free, irrevocable license to SM-102-based LNP technology. mCOMBRIAX EU approval therefore does not activate an ongoing royalty stream for Arbutus/Genevant; their economic benefit from mCOMBRIAX is captured entirely in the lump-sum settlement proceeds. The commercial significance of the mCOMBRIAX CHMP positive opinion for Arbutus/Genevant is retrospective — it validates the asset value underpinning the settlement amount — not prospective.
EMA CHMP Opinions — February 27, 2026 (Reported and Acted Upon During W10)
The CHMP meeting of February 23–26 produced positive opinions published February 27 that entered regulatory and market workflows during the March 2–7 window. Key items beyond the already-tracked Accord/Foresee CAMCEVI 21mg:
| Product | Active Substance | Indication / Notes |
|---|---|---|
| Moderna mCOMBRIAX | COVID-19/influenza mRNA combination | First combined COVID-19/influenza mRNA vaccine globally; positive opinion for adults ≥50; ahead of FDA on this combination |
| Ipsen Ojemda | Tovorafenib | Conditional MA for pediatric low-grade glioma (ages ≥6 months) |
| Palsonify | Paltusotine | Positive opinion for acromegaly (oral somatostatin receptor ligand) |
| Rhapsido | Remibrutinib | Positive opinion for chronic spontaneous urticaria (BTK inhibitor) |
| X4 Pharmaceuticals Xolremdi | Mavorixafor | Exceptional circumstances authorization for ultra-rare WHIM syndrome (ages ≥12) |
| Onerji | Levodopa/carbidopa | Positive opinion for advanced Parkinson's disease |
| Sanofi/DNDi acoziborole | Acoziborole | First single-dose oral treatment for human African trypanosomiasis (sleeping sickness) — approved under the EU-M4All procedure for use outside the EU; landmark neglected disease approval |
| Dupixent extension | Dupilumab | Pediatric chronic spontaneous urticaria, children aged 2–11 (first biologic for CSU under 12) |
| Jorveza extension | Budesonide | Pediatric eosinophilic esophagitis extension (ages 2–17) |
| Keytruda, Olumiant, Scemblix, Stelara | Various | Indication extensions |
Six biosimilars received positive opinions: Bysumlog (insulin lispro), Dazparda (insulin aspart), Fubelv (etanercept), Poherdy (pertuzumab), Tuyory (tocilizumab), and Zandoriah (teriparatide).
Negative opinions: Acadia's Daybu (trofinetide) for Rett syndrome received a negative opinion — notably, the product is already FDA-approved in the U.S. as Daybue. Vanda Pharmaceuticals' iloperidone for schizophrenia/bipolar disorder also received a negative opinion (its third EMA rejection). Withdrawal: Pfizer withdrew its sasanlimab (Zumrad) marketing application for bladder cancer following EMA concerns about mid-study protocol and statistical methodology changes.
Kedrion Biopharma | Commercial Launch / Distribution | Primary Humoral Immunodeficiency | Approved (FDA, late 2025)
Announced March 6, 2026, Kedrion Biopharma activated nationwide U.S. commercial distribution of QIVIGY (immune globulin intravenous, human-kthm), a 10% IVIG therapy approved by the FDA in late 2025 for the treatment of adults with primary humoral immunodeficiency (PI). QIVIGY is Kedrion's first fully proprietary IVIG — developed, manufactured, and released through the company's own integrated global network — representing more than a decade of internal R&D and manufacturing investment. Kedrion operates 68 plasma collection centers in the U.S. and 8 in the Czech Republic, with 7 production facilities across 5 countries.
The U.S. distribution network covers specialty pharmacies, infusion centers, hospitals, and other sites of care through the following named partners:
| Partner | Role |
|---|---|
| CuraScript SD | Specialty distributor |
| Cencora, Inc. | Specialty distributor |
| Cardinal Health, Inc. | Specialty distributor |
| McKesson Plasma and Biologics | Specialty distributor |
| BioCare, Inc. | Specialty distributor |
| Premier, Inc. | Exclusive GPO (acute and non-acute care) |
| Premier Innovatix | Exclusive GPO (acute and non-acute care) |
No financial terms disclosed. This is a commercial launch announcement, not a licensing or M&A transaction. QIVIGY is indicated for adults with primary humoral immunodeficiency — a category of rare inherited conditions where patients cannot produce sufficient antibodies, requiring lifelong immunoglobulin replacement therapy.
The IVIG market is large, competitive, and supply-constrained, with Kedrion positioning QIVIGY on the basis of integrated manufacturing control and supply reliability rather than clinical differentiation.
Asia-Pacific Regulatory and Market Events
Japan: World-First iPS Cell Therapy Approvals — March 6, 2026
The MHLW formally approved two iPSC-derived products on March 6, 2026 under Japan's conditional/time-limited approval pathway — a landmark moment in regenerative medicine nearly 20 years after Yamanaka's 2006 discovery of induced pluripotent stem cells.
| Product | Developer | Cell Type | Indication | Data Requirement |
|---|---|---|---|---|
| ReHeart | Cuorips | iPSC-derived cardiomyocyte sheets (~100M cells) | Ischaemic cardiomyopathy | 75 patients, 7 years |
| Amchepry | Sumitomo Pharma | iPSC-derived dopaminergic neural progenitors (5–10M cells) | Parkinson's disease | 35 patients, 7 years |
ReHeart consists of approximately 100 million iPSC-derived cardiomyocyte sheets transplanted onto the heart surface for ischaemic cardiomyopathy. Market availability is expected in autumn 2026. Sumitomo Pharma's Amchepry delivers iPSC-derived dopaminergic neural progenitor cells for Parkinson's disease; a trial of 7 patients showed symptom improvement in 4, with no major adverse effects. Neither trial included a control group. Both approvals carry a 7-year post-marketing evidence generation requirement before full approval can be considered.
China: Sciwind ecnoglutide (Severwin / Xianweiying) — NMPA Obesity Approval
Sciwind Biosciences / Pfizer China — ecnoglutide | NMPA Approval | Obesity | Marketed | March 6, 2026
| Parameter | Detail |
|---|---|
| Approved | March 6, 2026 (NMPA) |
| Asset | Ecnoglutide (brand names: Severwin, Xianweiying) |
| Developer | Sciwind Biosciences |
| China commercialization | Pfizer (exclusive rights secured February 24, 2026, for up to $495M) |
| Indication | Chronic weight management in adults with BMI ≥28, or ≥24 with comorbidities |
| Phase 3 data | 15.4% mean weight loss at 48 weeks (15.1% placebo-adjusted); 63.5% of patients achieved ≥15% weight loss |
| Mechanism | First cAMP-biased GLP-1 agonist approved for weight management globally |
| Designation | Pfizer's first obesity drug approval |
Japan: Shionogi Xocova — COVID-19 Prevention Indication Panel Approval
The MHLW expert panel recommended adding a prophylaxis indication to Xocova (ensitrelvir), Shionogi's approved oral COVID-19 treatment. If ministerially approved, Xocova would become the first oral COVID-19 prevention treatment in Japan.
Japan: Daiichi Sankyo MMR Vaccine — Expert Panel Approval
The MHLW panel greenlit ministerial approval of Japan's first MMR vaccine in over three decades. Japan suspended all MMR vaccination in 1993 due to safety concerns; this approval, primarily indicated for children aged 12 months and older, marks the restoration of combined measles-mumps-rubella immunization.
Japan: Takeda oveporexton — NDA Filed for Narcolepsy Type 1
Takeda filed an NDA with the MHLW for oveporexton for narcolepsy type 1. Oveporexton is a first-in-class orexin receptor 2-selective agonist targeting the root neurochemical deficit of narcolepsy. Also in late-stage development at Idorsia (under its own NDA in other jurisdictions).
South Korea: Nxera Pharma daridorexant — MAA Filed with MFDS
Nxera Pharma filed a marketing authorization application with Korea's Ministry of Food and Drug Safety for daridorexant (QUVIVIQ), a dual orexin receptor antagonist for insomnia. Daridorexant is already approved in Japan (via Shionogi), the US/EU (Idorsia), and China (Simcere). The Korea filing extends the product's geographic footprint.
India: Semaglutide Generic Market Opening — 50+ Brands Preparing for Launch
Composition and delivery patents for semaglutide expired in India during March 2026, opening the market to generic competition. More than 50 generic semaglutide brands are in preparation for launch from Indian manufacturers including Natco Pharma, Dr. Reddy's, Sun Pharma, Zydus, Mankind Pharma, and others. Natco Pharma confirmed a March 2026 launch. The Indian semaglutide generic market is expected to represent a significant reduction in treatment costs domestically and establish Indian manufacturers as generic semaglutide suppliers for lower-income markets globally.
India-EU Free Trade Agreement — Pharmaceutical Tariff Implications
The India-European Union Free Trade Agreement — described as the largest FTA concluded by either side — was concluded in early March 2026. The agreement phases out India's 11% pharmaceutical tariff over 5–10 years and opens reciprocal access to the EU's pharmaceutical and medtech market. The implications for the pharmaceutical industry are broad: Indian generic manufacturers gain improved access to EU markets, while European innovators gain improved terms for originator product access into India.
Additional Transactions
The following transactions were announced during the week. Entries vary in analytical depth; some include summary context only.
Azenta / UK Biocentre — GBP 20.5M Acquisition | Acquisition | Life Science Sample Management | Commercial | March 4, 2026
| Parameter | Detail |
|---|---|
| Deal value | GBP 20.5M net of cash (incl. up to GBP 1.8M contingent consideration upon milestone completion) |
| USD equivalent | ~$27.4M |
| Target trailing revenue | ~GBP 15.3M (12 months ended September 30, 2025) |
| Purchase multiple | Not disclosed; revenue multiple implied ~1.3x TTM |
| Acquirer | Azenta, Inc. (Nasdaq: AZTA), via wholly owned subsidiary Azenta UK Ltd |
| Target | UK Biocentre Limited (founded 2014; Hertfordshire, UK) |
| Financial advisor | Deloitte (Azenta) |
| Legal advisor | Bird & Bird LLP (Azenta) |
| Financial impact | ~35bps dilutive to 2026 Adjusted EBITDA margin; expected accretive to 2027–2028 organic revenue growth and margin expansion targets |
| Planned deployment | BioArc Ultra (16M-sample automated biorepository) to be installed at UK Biocentre site later in 2026 |
| Transaction webcast | March 10, 2026 |
| Announced / completed | March 4, 2026 |
Azenta completed the acquisition of UK Biocentre Limited on March 4, expanding its Sample Repository Services (SRS) business in the United Kingdom and establishing UK Biocentre as a European-wide operational hub. UK Biocentre provides end-to-end sample management, storage, and high-throughput processing for large-scale biomedical, pharmaceutical, and clinical studies, generating approximately GBP 15.3M in trailing revenue and supporting population-scale programs including academic, pharma, and public health clients. The company will continue to operate under its existing brand.
The acquisition complements Azenta's existing European biorepository in Griesheim, Germany and positions the combined infrastructure to serve multi-regional demand from pharmaceutical and biotechnology companies. Azenta plans to deploy its BioArc Ultra — a 16-million-sample automated cold-chain platform — at the UK Biocentre site, upgrading the facility's technological capabilities. Management guided modest near-term margin dilution (~35bps in 2026) offset by accretion to its publicly stated 2027–2028 organic growth targets.
Frontage Laboratories / Teddy Lab — Acquisition Completed | Acquisition | CRO/Central Laboratory Services (China) | Commercial | March 4–5, 2026
| Parameter | Detail |
|---|---|
| Target | Teddy Clinical Research Laboratory (Shanghai), Ltd. ("Teddy Lab") |
| Acquirer | Frontage Laboratories, Inc. (subsidiary of Frontage Holdings Corporation, HKEX: 1521.HK) |
| Financial terms | Not disclosed |
| Teddy Lab credentials | CAP-accredited, NGSP Level 1 certified; Class II Unsealed Radioactive Material Workplace; 1,500+ clinical trials supported; contributed to 50 approved drugs |
| Geographic rationale | Expands Frontage's central laboratory footprint in China for domestic and multi-regional clinical trials (MRCT) |
| Announced / completed | March 4 (announced), March 5 (PR Newswire distribution) |
Frontage Laboratories announced the completed acquisition of Teddy Clinical Research Laboratory (Shanghai) on March 4–5, extending its China-based central laboratory capacity. Teddy Lab holds internationally recognized quality accreditations (CAP, NGSP Level 1) and has supported more than 1,500 clinical trials across therapeutic areas, contributing to the regulatory approval of 50 new drugs. No financial terms were disclosed.
Frontage — a wholly owned subsidiary of Frontage Holdings (HKEX: 1521.HK, market cap ~$285M, TTM revenue ~$253M) — operates 26 sites globally as a full-service CRO/CDMO offering discovery through late-phase clinical services. The Teddy Lab integration is expected to support growing demand for domestic China clinical trials and multi-regional studies as international sponsors increasingly run bridging studies across Asian markets. The acquisition adds central lab capabilities to Frontage's existing bioanalytical, DMPK, safety/toxicology, CMC, and clinical trial services, reinforcing its one-stop-shop positioning.
Guangzhou Baiyunshan Pharma / Zhejiang Medical Industrial — RMB 500.5M Acquisition Completed | Completed March 3, 2026 (originally announced December 17, 2025)
Guangzhou Pharmaceutical Co. (广州医药, 874839), the core commercial subsidiary of Guangzhou Baiyunshan Medical Group (SSE: 600332), completed its acquisition of 100% of Zhejiang Provincial Medical Industrial Co. (浙江省医药工业有限公司, "Zhejiang Yigong") from Zhejiang Hisun Pharmaceutical (SSE: 600267) for RMB 500.5M (~$69M). The transaction was originally agreed December 10–17, 2025 via a public listing process on the Taizhou Property Exchange, with Guangzhou Pharma winning the bid at RMB 500.5M and paying an RMB 86M deposit. Chinese antitrust clearance (SAMR) was required and obtained; completion and business registration transfer were confirmed in the March 3 announcement.
Zhejiang Yigong holds specialist distribution licenses including controlled substances (ephedrine, caffeine, Schedule II narcotics) and has a province-wide hospital and retail distribution network, which extends Guangzhou Pharma's reach into the Yangtze River Delta. No advisors disclosed. Reported via SSE regulatory filing and Chinese financial press only; no international media coverage.
Fagron NV / Vepakum — Brazilian Pharmaceutical Packaging Acquisition Completed | Completed March 3, 2026 (originally announced December 2025)
Fagron (Euronext Brussels/Amsterdam: FAGR), the Belgian-Dutch global pharmaceutical compounding leader operating in 38+ countries, announced completion of its acquisition of Vepakum, a Brazilian pharmaceutical packaging company operating two facilities in São Paulo. The deal was originally announced in December 2025; Brazilian antitrust clearance from CADE was obtained in early February 2026, enabling the March 3 closing. Vepakum specializes in high-quality pharmaceutical packaging solutions and marks Fagron's entry into a new vertical — packaging — intended to generate operational synergies through integrated packaging, distribution, and shared services within its Latin American compounding network.
No transaction value was disclosed. This is one of several simultaneous Fagron acquisitions; the acquisitions of Injeplast and Amber remain pending close. Reported via Fagron regulated disclosure (GlobeNewswire) only; no Reuters or Bloomberg coverage.
Alibaba Health / Meinian Onehealth — Home Nursing Strategic Alliance | March 2, 2026
Alibaba Health (HKEX: 241) and Meinian Onehealth Healthcare Group (SZSE: 002044) signed a strategic cooperation agreement on March 2, 2026 covering three pillars: home nursing service coordination, online-offline resource integration, and pharmacy-health linkage. The operational model pairs Alibaba Health's digital platform and user traffic against Meinian's network of approximately 600 physical health screening centers nationwide, which serve as dispatch hubs, nurse workstations, and supply storage points for home visits. The collaboration aims to build a "30-minute health service circle" offering chronic disease management, post-operative care, and rehabilitation nursing at home, in line with China's national "Internet+ Nursing Services" policy push.
A joint insurance product combining preventive screening and health coverage is also planned. No financial terms disclosed. Reported only in Chinese financial press (Sina Finance, Securities Times); no Reuters or Bloomberg coverage found.
BARDA / Orexo US — OX390 Development Award, Up to $50.9M | Originally announced September 29, 2025; referenced in W10 Orexo/Dexcel context
Note: This award predates the W10 reporting window (March 1–6, 2026). It is included here because it is referenced in the Dexcel Pharma / Orexo Zubsolv entry as part of Orexo's post-divestiture strategic context.
The U.S. Biomedical Advanced Research and Development Authority (BARDA), part of the Administration for Strategic Preparedness and Response within HHS, awarded Orexo US, Inc. $8M in initial funding for the development of OX390, an intranasal rescue medication for adulterated opioid overdoses. The award is structured in five milestone-gated stages and is valued at up to $50.9M if all stages are completed through regulatory filing.
Orexo retains all commercial rights to OX390 upon FDA approval.
OX390 is a new chemical entity (NCE) designed to reverse respiratory depression in overdoses involving opioids adulterated with xylazine or medetomidine — alpha-2 adrenergic agonists (veterinary tranquilizers) that are increasingly common adulterants in the illicit drug supply and that standard opioid antagonists such as naloxone do not adequately reverse. The product uses Orexo's AmorphOX dry-powder intranasal delivery platform, which provides rapid absorption without the freezing risk of liquid nasal sprays — a practical advantage for community-based and outdoor first-responder use. OX390 is currently in preclinical/IND-enabling stage.
The contract number is 75A50125C00010.
Companion Spine / Xtant Medical — $21.4M total, $10.7M final payment | March 2, 2026
Xtant Medical (NYSE American: XTNT) received a final $10.7M payment from Companion Spine, completing the divestiture of its Coflex and CoFix interlaminar stabilization implant assets and all Paradigm OUS businesses. The deal originated in December 2025 and brings total proceeds to $21.4M, structured as an initial deposit plus promissory note ($8.2M) and working capital adjustments ($2.2M). Coflex is an FDA PMA-approved posterior motion preservation device with 90% patient satisfaction data across 150+ publications and over 12 years of U.S. commercialization.
Companion Spine, backed by Viscogliosi Brothers, is building what it describes as the largest posterior and cervical dynamic stabilization franchise globally. For Xtant, the divestiture exits implant hardware to focus on higher-margin orthobiologics.
Advisors: Companion Spine — Dorsey & Whitney LLP (lead legal), McDermott Will & Schulte (tax and German legal), Gide Loyrette Nouel (French legal), Farber LLC (IP legal), Natixis Partners (financial), FTI Consulting (strategic communications), Deloitte Finance (financial due diligence), MCRA (regulatory/clinical/reimbursement); Xtant Medical — Fox Rothschild LLP (legal).
Global Intelligence
Latin America: Sanofi Exits Brazilian Generics, GLP-1 Patent Cliff Spreads
EMS/NC Group — Sanofi Medley Acquisition | $600M | March 6, 2026
EMS/NC Group signed a definitive agreement to acquire Medley from Sanofi for $600M (R$3.175 billion) on March 6. Six bidders competed in the final round — Aché, Biolab, EMS, Hypera, Sun Pharma, and a Vinci Compass fund. EMS's geographic proximity, with Medley's Campinas factory situated approximately 20km from EMS's Hortolândia industrial park, was reported as decisive. The acquisition makes NC Group a ~R$12 billion revenue company and consolidates its position in Brazil's generics market. Subject to CADE antitrust approval. Lazard advised Sanofi; Mayer Brown advised EMS.
This is the largest single pharma M&A transaction in Brazil in several years and represents Sanofi's full exit from the Brazilian generics business, following a broader global strategy of divesting mature generics assets to focus on specialty and biologics.
Brazil — Novo Nordisk Semaglutide Patent Expiry | March 2026
Novo Nordisk's core semaglutide patent expired in Brazil in March 2026 following the Federal Supreme Court's denial of an extension request. The expiry opens competition in one of the world's largest GLP-1 markets, estimated at a ~$580M total addressable market. Hypera, Biomm (partnered with Biocon), EMS, Eurofarma, and Aché are all positioning for generic launches. EMS invested over R$1 billion in local peptide production and launched its first GLP-1 pen in August 2025.
ANVISA — Foreign Trade Health Control Regulation Update | March 4, 2026
ANVISA approved updated foreign trade health control regulations at its March 4 meeting, including results from international inspection missions to India and South Korea and expanded technical cooperation agreements.
Africa: ARV Procurement, Lenacapavir Access, and GLP-1 Positioning
Nigeria — ₦9.99B ARV Procurement Approval | March 5, 2026
Nigeria's Federal Executive Council approved ₦9.99 billion (~$6.24M) for Fidson Healthcare to procure antiretroviral drugs and build domestic ARV manufacturing capacity, addressing concerns about potential HIV medication shortages following PEPFAR uncertainty. FEC also approved equipment upgrades at six national cancer treatment centers.
South Africa — Lenacapavir Local Manufacturing Gap | March 6, 2026
South Africa is working with Unitaid and the US Pharmacopeia to identify a local manufacturer for Gilead's lenacapavir. Gilead's existing six voluntary licenses cover manufacturers in India, Egypt, and Pakistan, explicitly excluding South African firms — a gap drawing criticism from access advocates. Aspen Pharmacare is identified as a potential candidate.
Aspen Pharmacare — H1 FY2026 Results + GLP-1 Strategy | March 3, 2026
Aspen Pharmacare reported H1 FY2026 results showing revenue declined 4% to R21.09 billion, with profit dropping 38% due to R695 million in sterile manufacturing restructuring costs. CEO Stephen Saad announced plans to register Eli Lilly's Mounjaro (tirzepatide) across sub-Saharan Africa, targeting over 1 billion rand (~$61M) in 2026 sales. Aspen is also pursuing Health Canada approval for generic semaglutide between May–September 2026, which would serve as a regulatory benchmark for Latin American and Middle Eastern markets.
African Development Fund — $9.57M SADC Health Grant | March 3, 2026
The African Development Fund approved a $9.57M grant for the Resilient Health Systems for Emergency Preparedness Project across SADC nations, including upgrading Mozambique's Instituto Nacional de Saúde and strengthening Lesotho's national blood bank.
DRC / US — $1.2B Bilateral Health Partnership | March 2, 2026
The Democratic Republic of Congo and the United States sealed a $1.2 billion bilateral health partnership on March 2, one of the largest bilateral health commitments in DRC history.
Pan-African Climate and Health Consortium — £60M Launch | March 5, 2026
A Pan-African Climate and Health Consortium backed by approximately £60 million (~$75M) was launched on March 5 by the University of the Witwatersrand and Kwame Nkrumah University, supported by the Africa CDC's climate-health framework.
Africa CDC / Japan Institute for Health Security — Memorandum of Cooperation | March 5, 2026
Africa CDC and Japan's Institute for Health Security signed a Memorandum of Cooperation on March 5 covering infectious disease surveillance, laboratory capacity building, pandemic preparedness, and antimicrobial resistance. The agreement formalizes bilateral collaboration on public health security between the two organizations.
US — Bilateral Health MOUs with Central American and Caribbean Nations | March 2–7, 2026
The United States signed bilateral health memoranda of understanding with the Dominican Republic, El Salvador, Guatemala, and Panama during the week, focused on disease surveillance and health security under the broader "America First" bilateral health program. The simultaneous execution across four countries reflects the administration's shift toward direct bilateral health partnerships in place of multilateral global health frameworks.
India: Generics Leadership and Regulatory Development
Cipla / Kemwell Biopharma — Biologics Joint Venture | March 2, 2026
Cipla signed a joint venture with Kemwell Biopharma to develop, manufacture, and commercialize biologic products for global markets. Cipla holds 60% equity; Kemwell holds 40%, with initial investment up to ₹10 crore. Kemwell operates a US FDA-certified biologics facility in Bengaluru with 5,000-litre bioreactor capacity.
Emcure / Roche — India Nephrology Distribution | March 2, 2026
Emcure Pharmaceuticals signed a distribution agreement with Roche for nephrology and transplant products in India — specifically CellCept (immunosuppressant), Mircera, and Neorecormon (anemia therapies). Effective April 1, 2026.
Sun Pharma / Organon Acquisition Speculation | March 2–4, 2026
Sun Pharma was reported (Outlook Business, March 2) to have submitted a non-binding all-cash bid for Organon, with a valuation of approximately $10 billion including debt and $10–14 billion in financing arranged. Sun Pharma filed a stock exchange clarification on March 4 characterizing the reports as speculative. Unconfirmed. If completed, this would represent the largest overseas acquisition by an Indian pharmaceutical company.
Glenmark — First Generic Flovent HFA Approval | March 3–4, 2026
Glenmark received final FDA approval for fluticasone propionate inhalation aerosol 44 mcg, the first generic of GSK's Flovent HFA for asthma. Glenmark received Competitive Generic Therapy designation with 180 days of market exclusivity in a ~$520M annual US market.
India CDSCO — BA/BE Amendment Rules | March 7, 2026
India's CDSCO bioavailability/bioequivalence amendment rules took effect March 7, replacing approval requirements for low-risk studies with a prior-intimation mechanism. The change is expected to significantly accelerate generic drug development timelines.
Bharat Biotech — ~$500M IPO Plans | March 2, 2026
Bharat Biotech's approximately $500M IPO plans were reported on March 2. No formal filing has been made.
GCC and MENA
Cipla — Saudi Arabia Subsidiary Incorporation | March 1–2, 2026
Cipla incorporated a wholly owned subsidiary in Saudi Arabia (Cipla Middle East Company) for pharmaceutical manufacturing, distribution, and marketing authorization — a strategic entry into the $8+ billion Saudi generics market aligned with NUPCO localization requirements.
Insilico Medicine / Emirates Drug Establishment — AI Drug Discovery MoU | March 5–6, 2026
Insilico Medicine signed an MoU with the Emirates Drug Establishment to advance AI drug discovery tools in the UAE. Insilico shares rose 12.8% on the announcement. The company also plans to establish a Riyadh discovery cell in 2026, backed by Prosperity7 Ventures (Aramco).
Hikma Pharmaceuticals — Buyback Continuation, CEO Change | March 4, 2026
Hikma Pharmaceuticals continued executing its $250M share buyback, purchasing 244,980 shares at 1,254.07p on March 4. The program followed major leadership changes: CEO Riad Mishlawi departed and Executive Chairman Said Darwazah assumed CEO responsibilities. Hikma stock had declined ~16% on weak 2026 guidance of 2–4% growth against a 5.6% consensus estimate.
Egypt — National Pharmaceutical Export Roadmap | March 2, 2026
Egypt launched a national pharmaceutical industry roadmap targeting $3 billion in annual medical exports, including manufacturing localization, AI/biotech adoption, and concessional financing raised to EGP 150 million credit limits.
Europe: Bayer Settlement, Novo Nordisk Dual Moves, Lonza Divestiture
Bayer — $7.25B Roundup Settlement Preliminary Court Approval | March 4, 2026
A Missouri circuit court granted preliminary approval on March 4 for Bayer's $7.25 billion Roundup class-action settlement covering approximately 65,000 remaining NHL lawsuits. Court: 22nd Judicial Circuit Court, City of St. Louis, Missouri (Randall King, et al. v. Monsanto Company, Case No. 2622-CC00325). Presiding judge: Timothy Boyer. Settlement administrator: BrownGreer PLC. Allocation Special Master: Matt Garretson. Official settlement website: WeedKillerClass.com. Payment structure: $500M within 10 business days of preliminary approval, another $500M by August 31, 2026, followed by 16 annual payments declining from $550M to $250M with up to 2.5% annual inflation adjustment from year 5, plus a separate $1B security fund as bankruptcy backstop. The 90-day opt-out period runs until June 4 — critically, before the Supreme Court is expected to rule on Monsanto Co. v. Durnell (No. 24-1068, oral arguments April 27, 2026). Bayer's CEO Bill Anderson stated they need participation "very close to 100%." Fourteen law firms representing ~20,000 plaintiffs filed motions to intervene. Fairness hearing set for July 9. Individual payouts range from $10,000–$165,000+ based on age, exposure type, and NHL severity. Lead class counsel: Christopher Seeger (Seeger Weiss). Total Roundup litigation costs now exceed $11 billion. Bayer reported FY2025 sales of €45.6B with a net loss of €3.6B from litigation charges.
Novo Nordisk — €432M Irish Factory Expansion + Hims & Hers Platform Deal | March 2–7, 2026
Novo Nordisk announced a €432M (~$507M) investment on March 2 to upgrade its tabletting facility in Monksland, Ireland for oral GLP-1 manufacturing serving markets outside the US. Construction is underway with completion expected through 2027–2028. Separately, on March 7, Bloomberg reported that Novo Nordisk will sell Wegovy on the Hims & Hers platform starting at $599/month, ending a public legal dispute between the companies. Hims & Hers shares surged approximately 40% in after-hours trading on the news. The Hims deal is directly relevant to the royalty market: it resolves uncertainty about branded GLP-1 channel strategy and supports the royalty cash flow trajectory for royalty holders on Novo's GLP-1 franchise.
Lonza — Personalized Medicine Business Divestiture | March 4, 2026
Lonza signed a definitive agreement on March 4 to divest its Personalized Medicine business — including the Cocoon automated cell therapy production platform — to Canada's Octane Medical Group. The divestiture reflects Lonza's strategic focus on its core CDMO operations. Expected to close by end of Q1 2026.
Merck KGaA — FY2025 Results, SpringWorks Pipeline Write-off, CEO Succession | March 5, 2026
Merck KGaA reported FY2025 net sales of €21.1 billion and guided 2026 revenue of €20.0–21.1B, assuming no U.S. Mavenclad sales from March 2026 due to generic entry following patent invalidation. Mavenclad posted €1.19B in 2025 sales (+17%) before the patent cliff. The stock fell approximately 8% on the results day.
The most significant buried item in the earnings disclosure: Merck KGaA confirmed it has discontinued all clinical-stage oncology candidates acquired through its $3.9 billion SpringWorks Therapeutics acquisition — including SW-682 (Hippo/TEAD pathway inhibitor) and M9466 (PARP1-selective inhibitor) — retaining only the two commercially approved drugs Ogsiveo (nirogacestat) and Gomekli (mirdametinib). This represents one of the largest pipeline write-offs from a single acquisition in recent European pharma history and materially re-prices the strategic rationale for the SpringWorks deal. The discontinuations do not affect any currently disclosed royalty streams but are relevant as a signal of disciplined pipeline rationalization under incoming leadership.
CEO Belén Garijo's planned departure was confirmed, with Kai Beckmann named as successor effective later in 2026. Separately, Garijo was confirmed as incoming CEO of Sanofi — a high-profile executive move that simultaneously reshapes two major European pharma boardrooms.
Novartis AGM — 29th Consecutive Dividend Increase | March 4–7, 2026
Novartis held its AGM, approving a 5.7% dividend increase to CHF 3.70/share — its 29th consecutive annual increase. The $12B Avidity Biosciences acquisition remains on track for H1 2026 closure.
Galapagos — 1.75M Employee Subscription Rights Created | March 6, 2026
Galapagos created 1,750,000 subscription rights under its 2026 employee stock plan on March 6, as part of routine equity compensation administration.
Genmab — Share Buyback Continuation | March 2–7, 2026
Genmab continued executing its share buyback program initiated February 17, 2026, purchasing shares throughout the week in open market transactions consistent with the company's stated capital return policy.
UK Regulatory Activity | March 4–5, 2026
Ananda Pharma received MHRA approval for its ENDOCAN Phase 2 trial of MRX1 CBD solution for endometriosis pain. Zelluna Therapeutics received MHRA CTA approval for ZIMA-101, a first-in-human TCR-NK cell therapy trial at The Christie NHS Foundation Trust. The UK Space Agency and MHRA issued a joint statement supporting in-orbit pharmaceutical manufacturing.
APAC: Japan Price Cuts, South Korea Emerges as Deal Hub, SK Plasma Technology Transfer
Japan — FY2026 NHI Drug Price Revision | March 5–7, 2026
Japan published its FY2026 NHI drug price revision implementing a 4.02% average price cut saving ¥91.8 billion. For the first time, the G1 price cut rule applies to originator biologics, with up to 13 brands affected. Notable cuts include Avastin at 45% and Forxiga at 36%. The revision takes effect April 1. For royalty holders on Japan-commercialized assets, the revision reduces the revenue base against which royalties are calculated and should be incorporated into cashflow models from Q2 2026.
Japan — MHLW New Molecular Entity Approvals | March 5–6, 2026
Japan's MHLW panel recommended approval of three new molecular entities: OrphanPacific's leniolisib (Joenja) for activated PI3Kδ syndrome, Ultragenyx Japan's triheptanoin (Dojolvi) for fatty acid oxidation disorders, and Bayer's gadoquatrane (MRI contrast agent). Label expansions for Lusefi and Effexor were also recommended.
Japan — Shionogi Xocova COVID-19 Prevention Indication | March 2026
The MHLW expert panel recommended adding a prophylaxis indication to Xocova (ensitrelvir). If ministerially approved, Xocova would become the first oral COVID-19 prevention treatment in Japan.
Japan — Daiichi Sankyo MMR Vaccine Panel Approval | March 2026
The MHLW panel approved Japan's first MMR vaccine in over three decades. Japan had suspended combined MMR vaccination in 1993; this approval restores combined measles-mumps-rubella immunization primarily for children aged 12 months and older.
Japan — Takeda Oveporexton NDA Filed for Narcolepsy | March 2026
Takeda filed an NDA with the MHLW for oveporexton for narcolepsy type 1. Oveporexton is a first-in-class orexin receptor 2-selective agonist targeting the root neurochemical deficit of narcolepsy.
South Korea — K-health MIRAE Initiative | $110M | March 5, 2026
South Korea's K-health MIRAE initiative — the country's ARPA-H equivalent — announced a ₩162 billion (~$110M) funding plan across 9 projects over 5 years focused on national health security, with project recruitment beginning May 2026.
SK Plasma / Proturk — €65M (~$75M) Technology Transfer | March 5, 2026
SK Plasma signed a €65M technology transfer deal with Turkey's Proturk on March 5 — the largest single contract since SK Plasma's 2015 founding. SK Plasma will license plasma fractionation technology for a 600,000 liters/year plant in Ankara and acquire a 15% equity stake. Turkey currently imports 100% of its plasma-derived products in a market estimated at approximately $500M annually.
South Korea — Cancer Drug Reimbursement Expansion | March 1, 2026
Korea expanded cancer drug reimbursement effective March 1, adding coverage for Imfinzi in hepatocellular carcinoma and bile duct cancer, Balversa in urothelial carcinoma, and Xpovio in multiple myeloma and cervical cancer.
Samsung Bioepis / Hanmi — Prolia Biosimilar Commercialization | March 2026
Samsung Bioepis partnered with Hanmi Pharmaceutical for Korean commercialization of Prolia biosimilar SB16 (denosumab), while Celltrion simultaneously cut the price of its competing denosumab biosimilar Stoboclo by 28%.
Daewoong — Nabota China NMPA Filing Accepted | March 4, 2026
Daewoong's Nabota (botulinum toxin) NMPA filing was accepted in China, reopening a regulatory pathway after voluntary withdrawal in July 2025.
China — Jiuyuan Genetic Semaglutide Biosimilar Filing | March 2026
Jiuyuan Genetic's semaglutide biosimilar for obesity was accepted for NMPA review — China's first semaglutide biosimilar application for weight management.
South Korea — Nxera Pharma Daridorexant MAA Filed | March 2026
Nxera Pharma filed a marketing authorization application with Korea's Ministry of Food and Drug Safety for daridorexant (QUVIVIQ), a dual orexin receptor antagonist for insomnia. Daridorexant is already approved in Japan, US/EU, and China.
Everest Medicines — NEFECON Full TFDA Approval in Taiwan | March 2026
Everest Medicines received full TFDA approval in Taiwan for NEFECON (budesonide delayed-release) for IgA nephropathy, broadening eligibility regardless of proteinuria level.
Roche / South Korea Ministry — KRW 710B ($481M) Five-Year Partnership
Note: The Roche/South Korea MOU ($481M, March 3, 2026) was captured in v7. The third sweep confirms South Korea as the week's most active single-country deal market outside the US, with the Roche MOU, SK Plasma/Proturk tech transfer, Samsung Bioepis/Hanmi partnership, K-health MIRAE initiative, expanded cancer reimbursement, and Daewoong's China filing occurring in parallel.
Southeast Asia: Cell Therapy, Infrastructure, and Access Partnerships
ImmunoScape — WT1-Positive Solid Tumor Trial MoU + DiCaprio Investment | March 4, 2026
Singapore-based ImmunoScape (A*STAR spin-out, backed by Amgen Ventures and EDBI) signed an MoU with an NCI-designated Cancer Center to launch a clinical trial targeting WT1-positive solid tumors across ovarian, mesothelioma, pancreatic, and colorectal cancers. The company also received a strategic investment from Leonardo DiCaprio.
Terumo BCT — Cell and Gene Therapy Ecosystem Blueprint | March 5, 2026
Terumo BCT launched its Cell and Gene Therapy Ecosystem Blueprint from Singapore — a collaborative framework to help APAC developers scale from early development to delivery, with partnerships with Precision Bio and Taiwan Bio Therapeutics.
Orbis / Roche Pharma Vietnam — Diabetic Blindness Prevention Partnership | March 5, 2026
Orbis and Roche Pharma Vietnam announced a two-year partnership to prevent avoidable blindness in diabetic patients, targeting approximately 27,000 patients across 7 hospitals in Hanoi and Ho Chi Minh City through AI-assisted screening.
KPJ Healthcare Berhad — Malaysia's First Neuroscience and Stroke Centre of Excellence | March 5, 2026
KPJ Healthcare Berhad launched Malaysia's first Neuroscience and Stroke Centre of Excellence on March 5 at Damansara Specialist Hospital 2, as part of its strategic roadmap to establish 15 Centres of Excellence by 2030. The launch is part of KPJ's hospital network expansion and specialty services strategy across Malaysia.
Global Health Financing: Global Fund, EDCTP3, and GLP-1 Access
Global Fund — GC8 Implementation Begins | March 4–5, 2026
WHO and the Global Fund held Grant Cycle 8 support webinars on March 4–5 to guide development of funding proposals for HIV, TB, and health systems. This follows the Global Fund Board's February 18 approval of $10.78B in country allocations for 2027–2029 plus $260M for catalytic investments.
EU EDCTP3 — €147M Research Call Deadline | March 4, 2026
The EU's EDCTP3 program had its March 4 deadline for the first stage of its €147M 2026 research funding call covering TB drugs, lower respiratory infections, HIV prevention, and climate-driven infectious diseases.
Medicines Patent Pool — GLP-1 Access Advocacy | March 4, 2026
The Medicines Patent Pool used World Obesity Day (March 4) to advocate for voluntary GLP-1 licenses in low- and middle-income countries, with Coefficient Giving joining as a new philanthropic funder supporting NCD medicines access.
Hiray Pharma Solutions — Orforglipron Manufacturing Milestone | March 4, 2026
Hiray Pharma Solutions (Jingmen, China) disclosed significant technical milestones in process optimization for orforglipron (Eli Lilly's oral non-peptidic GLP-1 agonist) manufacturing, integrating frontier organometallic chemistry — a notable CDMO capability signal ahead of what may become a highly competitive oral GLP-1 supply chain.
Securities Litigation
Pomerantz LLP / Mereo BioPharma Group (NASDAQ: MREO) and Ultragenyx Pharmaceutical (NASDAQ: RARE) | Securities Class Action | Osteogenesis Imperfecta | Phase 3 failure
Pomerantz LLP has filed class action lawsuits against both Mereo BioPharma and Ultragenyx Pharmaceutical following the December 29, 2025 announcement that both the Orbit and Cosmic Phase 3 studies for setrusumab (UX143) failed to achieve statistical significance on primary endpoints. Mereo's ADR price fell 42.5% on July 10, 2025 following a prior disclosure that the Orbit trial final analysis was being delayed; the subsequent Phase 3 failure compounded investor losses. The lead plaintiff deadline for both actions is April 6, 2026.
The litigation does not affect the underlying assets or create new deal activity, but is included here as a material legal development affecting two publicly traded biopharma counterparties whose royalty and milestone economics are relevant to the pharmaceutical royalty market. Ultragenyx held co-development rights and bore a substantial share of the program's late-stage costs.
Robbins LLP / Nektar Therapeutics (NASDAQ: NKTR) | Securities Class Action | Alopecia Areata | Phase 2b failure
Robbins LLP filed a securities class action on behalf of investors who acquired Nektar Therapeutics shares between February 26, 2025 and December 15, 2025. The complaint alleges that Nektar failed to disclose that enrollment in the REZOLVE-AA Phase 2b trial — evaluating rezpegaldesleukin (REZPEG, NKTR-358) in alopecia areata — had not followed applicable protocol standards, that this non-compliance was likely to materially impair the trial's results, and that public statements about the trial's integrity and prospects were accordingly misleading throughout the class period. On December 16, 2025, Nektar announced top-line results from the 36-week induction treatment period of REZOLVE-AA, disclosing that the trial failed to reach statistical significance and attributing the failure in part to the inclusion of four patients who should not have been eligible.
Nektar's stock fell $4.14 per share (7.77%) to close at $49.16 on that date. The lead plaintiff deadline is not yet disclosed in the public filing; shareholders wishing to serve as lead plaintiff should contact counsel. The announcement was filed March 6–7, 2026.
Context: Nektar's broader pipeline and investor dynamics. The REZOLVE-AA failure is the central litigation trigger, but the W10 disclosure environment around Nektar is materially more complex than the class action alone suggests. At the TD Cowen 46th Annual Health Care Conference on March 4, 2026, Nektar Chief R&D Officer Jonathan Zalevsky presented an updated clinical narrative that repositioned the company's immunology thesis around atopic dermatitis rather than alopecia areata. Zalevsky disclosed that Phase 3 initiation for REZPEG in atopic dermatitis is scheduled for next quarter (Q2 2026), with a second Phase 3 study to begin four to eight weeks thereafter.
The pivotal program is supported by 52-week maintenance data — reported in February 2026 — showing up to 85% maintenance of IGA response on quarterly dosing, with up to one in three patients achieving EASI-100 (complete skin clearance) at week 52, compared to approximately half of Dupixent patients losing IGA response on a monthly maintenance schedule. The company has now treated over 1,000 patients with REZPEG across multiple studies. Zalevsky also confirmed a Phase 2 collaboration with UCSF and neuroimmunologist Dr.
Stephen L. Hauser on NKTR-0165 (the company's TNFR2 agonist antibody) in multiple sclerosis studies, and a TrialNet-partnered trial in type 1 diabetes. Nektar executed a large equity raise following the 52-week AD data, substantially strengthening the balance sheet ahead of Phase 3 initiation.
On valuation, a Simply Wall St. analysis published March 3, 2026 noted that with NKTR last closing near $68 against an analyst consensus price target of $129.86, the stock trades at a 44.6% discount to the most widely followed fair value narrative — itself built on Phase 2b AD data and the near-$30B projected addressable market by 2033. On a price-to-sales basis, however, NKTR trades at 30.6x — far above the U.S. pharmaceuticals industry average of 4.2x and the comparable peer multiple of 12.7x — reflecting a pre-commercial premium that will compress or expand depending entirely on Phase 3 execution.
The Nektar situation is a layered risk-reward story that the securities litigation, taken in isolation, does not capture. The class action is backward-looking — REZOLVE-AA enrollment misconduct and a Phase 2b failure. The forward narrative is: dual Phase 3 initiation in atopic dermatitis in Q2 2026, with primary readout expected 2028, plus a new MS collaboration, an ongoing Type 1 diabetes program, and a strengthened balance sheet.
Whether the litigation overhang proves material to Phase 3 financing and partner development discussions depends on how quickly a lead plaintiff is designated and whether discovery surfaces additional disclosures not yet in the public record.
Report compiled March 7th, 2026. Transaction values represent announced figures; actual values may vary upon closing. Deals with undisclosed terms are noted but not included in aggregate value calculations. This report is for informational purposes only. The author is not a lawyer or financial adviser and this content does not constitute investment or legal advice.
Member discussion