The Royalty Swap: When Warrants Become Revenue Interests

When Immunic filed its 8-K on February 13, 2026, the headline was a $200 million private placement led by BVF Partners to fund the company's Phase 3 ENSURE trials in relapsing multiple sclerosis. Buried alongside it was a transaction that inverts the entire structural logic I described in The Warrant Sweetener: 51,087,000 Series B warrants, issued just eight months earlier in a $65 million public offering, were cancelled. In their place, holders received a pro rata share of a 5% synthetic royalty on future net sales of vidofludimus calcium. No cash changed hands. No new shares were issued. Equity exposure went in; product-linked revenue exposure came out.
In The Warrant Sweetener, I examined how warrants have become equity kickers within royalty restructurings, using ADC Therapeutics' deal with HealthCare Royalty as the primary case study. There, HCR received warrants from the company as consideration for loosening a $750 million change-of-control obligation. The Immunic transaction runs in the opposite direction. Warrant holders surrendered their equity instruments to the company and received royalty rights in return. If the ADC Therapeutics deal showed that royalty investors are acquiring equity optionality, the Immunic deal shows that equity investors are acquiring product-linked revenue streams. The boundary between the two asset classes is dissolving from both sides.
The Mechanics
Immunic's May 2025 public offering was structured as a triple-warrant package: pre-funded warrants, Series A warrants expiring December 31, 2025, and Series B warrants exercisable from October 1, 2025 through June 3, 2030. The combined offering price was $0.7499 per unit. Gross proceeds were approximately $65 million, with up to $130 million more if all warrants were exercised. The offering was co-led by BVF Partners and Coastlands Capital, with participation from Aberdeen Investments, Adage Capital Partners, and Janus Henderson Investors.
By February 2026, Immunic's stock was trading near $1.00. The company had received a Nasdaq delist warning for failing to maintain the minimum bid price and had transferred to the Nasdaq Capital Market. A reverse stock split of at least 10:1 was in preparation. Management had flagged substantial doubt about going concern in the Q3 2025 10-Q. The Series B warrants represented a massive equity overhang: 51 million potential shares on a base of roughly 120 million outstanding.
The Royalty Purchase Agreement, filed as Exhibit 10.3 to Immunic's 8-K on February 13, 2026, exchanged those warrants for royalty rights. A critical detail: the original Series B issuance comprised warrants to purchase up to 86,666,667 shares. Of these, holders of 51,087,000 warrants participated in the exchange, meaning roughly 59% of outstanding Series B warrants were tendered. The remaining 35,579,667 Series B warrants stayed outstanding.
Each Participating Series B Holder received a pro rata share of an aggregate 5% synthetic royalty on future sales of vidofludimus calcium "in any country," calculated as the number of warrants exchanged by that holder divided by the total warrants exchanged by all participating holders, expressed as a percentage. BVF Partners serves as royalty interest agent. Royalties are due and payable quarterly following the First Commercial Sale as defined in the agreement.
The 8-K also discloses that Immunic agreed to "specified affirmative and negative covenants, including without limitation covenants regarding periodic reporting of information by the Company to the Participating Series B Holders, and audits of royalties paid under the Royalty Purchase Agreement." The agreement contains representations and warranties, indemnification obligations, and other provisions described as "customary for transactions of this nature."
Crucially, Section 5.13 of the Securities Purchase Agreement for the concurrent $200 million private placement reveals that the warrant-to-royalty exchange was not an independent creative solution but a contractual commitment to the new investors. The provision states that "the Company further covenants to use its best efforts to, following the Closing Date, consummate an exchange offer of the Series B Warrants for an aggregate 5% synthetic royalty on future sales of the Company's IMU-838." The PIPE investors required cap table cleanup as a condition of their investment. The royalty swap was structurally linked to the equity raise from inception.

Leerink Partners acted as lead placement agent for the $200 million private placement, alongside Stifel, Guggenheim Securities, William Blair, LifeSci Capital, B. Riley Securities, and Brookline Capital Markets.
The Royalty That Came Back
There is an irony embedded in this transaction that only becomes visible through the asset's history. Immunic acquired vidofludimus calcium from 4SC AG in September 2016 through an asset acquisition that included an upfront cash payment, future milestone payments, and a 4.4% royalty on net sales. In March 2021, Immunic settled that royalty obligation for $17.25 million, paid 50% in cash and 50% in Immunic common stock. CEO Daniel Vitt described the deal as providing "100% of the future sales potential of our lead asset." No further payment obligations remained between Immunic and 4SC.
For nearly five years, vidofludimus calcium was royalty-free. Then, in February 2026, Immunic voluntarily re-encumbered the asset with a 5% synthetic royalty — a rate higher than the one it had just bought out. The net royalty trajectory is 4.4% → 0% → 5%.

The economics, however, are fundamentally different. Immunic paid $17.25 million in real consideration (cash and equity) to eliminate the 4SC royalty. It paid zero cash to create the new one. The 4SC royalty was a legacy obligation from an asset acquisition; the synthetic royalty is a cap table management tool. One was a cost of doing business. The other is the price of survival financing.
This creates an interesting analytical lens for the royalty market. The $17.25 million 4SC settlement implied that Immunic, in 2021, valued the NPV of a 4.4% royalty on vidofludimus calcium at roughly that amount — a meaningful premium given the asset was only in Phase 2 at the time. Five years later, Immunic created a larger royalty (5% versus 4.4%) on a more de-risked asset (Phase 3 versus Phase 2) and received no cash consideration for it. The warrant holders who received the royalty rights will either earn substantially more than $17.25 million if the drug succeeds, or nothing at all.
Is This a First?
In researching the landscape of pharmaceutical financings, I have not found a publicly documented precedent for a direct warrant-to-royalty exchange in the life sciences sector. The structure is genuinely novel.
Covington & Burling's synthetic royalty studies, which have tracked every publicly disclosed synthetic royalty transaction of $25 million or more since 2019, explicitly noted in their 2025 report that synthetic royalty deals "do not normally include additional warrant or equity kicker components." The standard model flows in one direction: cash today in exchange for a royalty tomorrow. Gibson Dunn's Royalty Report documented $29.4 billion in life sciences royalty financings from 2020 to 2024, overwhelmingly structured as cash-for-revenue transactions. Goodwin's October 2025 analysis confirmed this trend, noting that synthetic royalty structures have gained prominence within the broader royalty financing market.
None of these sources describe a transaction where a royalty was created not through a cash investment but through the cancellation of equity warrants. The Immunic 8-K itself describes the agreement as containing provisions "customary for transactions of this nature," but the nature itself, a warrant-for-royalty exchange, appears to be without direct precedent in the pharmaceutical context.
The closest structural analogues exist outside pharmaceuticals. In oil and gas financing, overriding royalty interests have occasionally been conveyed alongside warrants as part of lending packages, but these are additive, not substitutive. One 2006 SEC filing showed Goldman Sachs receiving both a 5% overriding royalty interest and warrants as part of a drilling facility, both instruments flowing in the same direction as additional consideration.
The Immunic structure is different in kind. It converts one instrument into the other, using a royalty as the price for extinguishing an equity obligation. As I discussed in The Warrant Sweetener, the pharmaceutical financing toolkit has been expanding, with warrants appearing in royalty restructurings (ADC Therapeutics), debt exchanges (Karyopharm), and serial inducements (Moleculin Biotech). But all of those transactions added warrants to existing structures. Immunic is the first to use a royalty to remove them.
Why This Structure Works
The transaction solved three problems simultaneously, and the SEC filings reveal it was designed to do so from the outset. Section 5.13 of the Securities Purchase Agreement shows the $200 million PIPE was conditional on Immunic committing to execute the warrant exchange. The new investors would not deploy capital into a cap table burdened with 86.7 million Series B warrants.
For Immunic, it eliminated 51 million warrants from the cap table ahead of a planned reverse stock split and a $200 million private placement. Warrant overhang depresses stock prices, complicates new equity issuances, and creates accounting complexity. Every warrant outstanding is a potential share that dilutes existing holders. Removing them without triggering cash exercise (which the sub-$1.00 stock price made impractical) or paying cash consideration (which the company could not afford) required creative structuring. A royalty, unlike cash or shares, costs nothing until the product generates revenue.
For the warrant holders, the exchange substituted binary equity exposure for product-linked cash flows. The Series B warrants were deep out of the money if the stock stayed near $1.00, and their value depended on a reverse stock split creating an artificially higher per-share price. A 5% royalty on a potential multiple sclerosis drug with blockbuster aspirations offered a different risk-reward profile: zero value if vidofludimus calcium fails its Phase 3 trials, but uncapped upside tied to annual net sales if the drug reaches the market.
For BVF Partners, who led both the original May 2025 offering and the February 2026 private placement, the exchange consolidated their economic exposure. BVF's Schedule 13D filed February 20, 2026 disclosed beneficial ownership of 13.6 million shares (9.99% of common stock), plus 53.3 million 2026 pre-funded warrants and 53.3 million common warrants across affiliated vehicles. By converting Series B warrants into royalty interests, BVF maintained exposure to vidofludimus calcium's commercial success through two distinct channels: equity upside through its substantial warrant and share position, and revenue participation through the synthetic royalty. BVF principal R. Thorvald Nagel simultaneously joined Immunic's board as a Class III director.
The Directionality Problem
The core contribution of this transaction to the pharmaceutical financing playbook is directionality. As I mapped in The Warrant Sweetener, the standard flow runs from company to investor: companies issue warrants to sweeten royalty amendments, induce warrant exercises, or compensate noteholders for accepting restructuring haircuts. The investor receives the equity kicker; the company receives the concession.
Immunic reversed the flow. Warrants moved from equity investors back to the company (via cancellation), and royalty rights moved from the company to those same investors.
The Karyopharm restructuring I examined in The Warrant Sweetener provides the middle case. There, convertible noteholders exchanged $148 million in principal for $111 million of new notes plus approximately 46 million warrants at $1.10. The warrants induced the exchange; without them, no bondholder would accept a 25% haircut on principal. But the warrants were equity instruments layered on top of a debt restructuring that also involved amending HCR's royalty rate on XPOVIO. In each case the instrument serves as the currency of negotiation. The difference lies in what it buys.
What 5% Buys on a Phase 3 MS Asset
Vidofludimus calcium targets relapsing multiple sclerosis through a dual mechanism: Nurr1 activation for neuroprotection and selective DHODH inhibition for inflammation reduction. The Phase 3 ENSURE trials are fully enrolled with top-line data expected by the end of 2026. If successful, Immunic plans a US NDA submission in mid-2027 with a targeted approval in 2028. The company is also initiating a Phase 3 programme in primary progressive MS (PPMS) in late 2026, estimated to take 3.5 to 4 years to complete — an indication expansion that would substantially broaden the royalty base.
The concurrent $200 million private placement included a structural trigger worth noting: the common warrants exercisable at $0.873 expire 30 days after the public announcement of ENSURE Phase 3 top-line data, or February 17, 2031, whichever is earlier. This creates a built-in forcing function: when the Phase 3 data drops, warrant holders have a narrow window to exercise or lose their equity position. A positive readout could drive rapid exercise of up to $200 million in additional capital.
The transaction also demonstrates synthetic royalty mechanics operating at a scale far below the institutional norm. At a market capitalisation of approximately $250 million, Immunic is a micro-cap — a far cry from the Royalty Pharma ($275 million to $2 billion range) or HealthCare Royalty deals that dominate the synthetic royalty landscape. This suggests the structure may be more broadly applicable than current market practice implies.
Immunic's management has described the global MS market as projected to reach over $30 billion by the early 2030s. GlobalData forecasts the MS DMT market across seven major markets at $25.9 billion by 2034, growing at a 1.7% CAGR. The broader market including emerging geographies is projected to reach approximately $38–39 billion by 2030–2033 according to Grand View Research and Mordor Intelligence.
Vidofludimus calcium is one of seven late-stage pipeline products that GlobalData identified as potentially reshaping the MS landscape, alongside four BTK inhibitors (Sanofi's tolebrutinib, Roche's fenebrutinib, Novartis' remibrutinib, and InnoCare's orelabrutinib) plus Sanofi's frexalimab and AB Science's masitinib. The oral therapy segment has established reference points for commercial success: Biogen's Tecfidera reached peak sales of approximately $4.4 billion before generic entry, Sanofi's Aubagio peaked around $2.0 billion, and Bristol Myers Squibb's Vumerity has reached approximately $1.0 billion.
Against these benchmarks, the scenario analysis for a 5% royalty is striking:
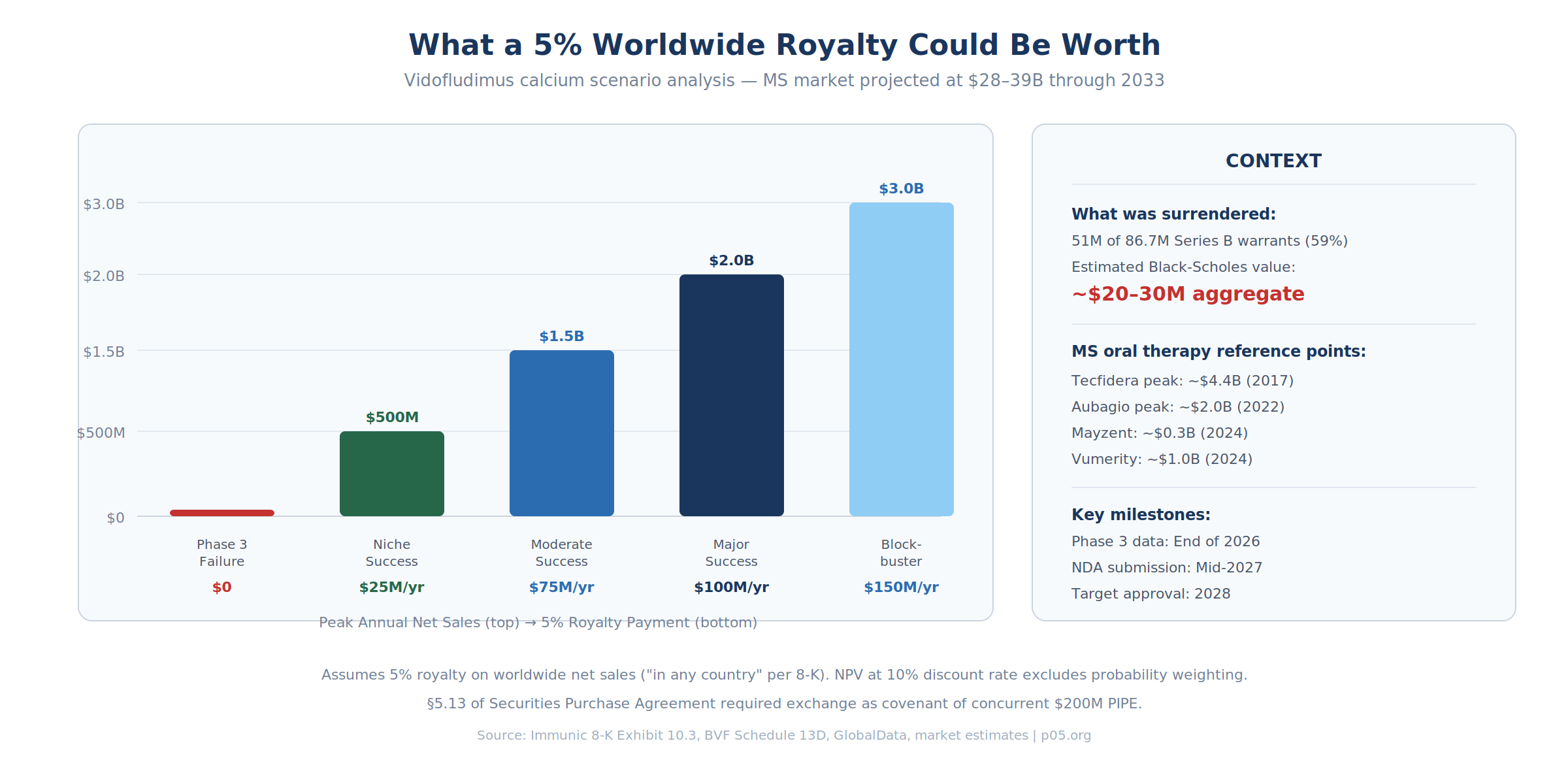
The 51 million cancelled warrants had a collective intrinsic value near zero at the time of exchange. Even assigning a generous time value using Black-Scholes with the stock at $1.00, five-year duration, and 80% implied volatility (typical for sub-$1 biotechs), the aggregate warrant value would be in the range of $20–30 million. The 5% royalty rate itself sits at the lower end of typical synthetic royalty financings, which generally range from 4–10% depending on stage and risk. The warrant holders accepted more modest royalty economics in exchange for eliminating the exercise-price risk inherent in the warrants — a rational trade when the underlying stock is near $1.00 and the company is fighting a Nasdaq delist warning. In any scenario where vidofludimus calcium achieves even niche commercial success, the royalty interest materially exceeds the surrendered warrant value. The asymmetry explains why the exchange worked: both sides could rationally believe they were getting the better deal.
Structural Questions
Several aspects of the Immunic transaction remain undisclosed despite the 8-K filing providing more detail than the initial press release. The filing confirms reporting covenants, audit rights, and indemnification obligations, but key questions persist.
Security interest. Post-Mallinckrodt, essentially every synthetic royalty financing includes a perfected security interest in the underlying intellectual property. Whether the Immunic royalty holders have a secured claim against the vidofludimus calcium patents and regulatory assets is not clear from the press release or 8-K summary. As I discussed in The Warrant Sweetener citing the Covington guide to synthetic royalty dealmaking, unsecured synthetic royalty holders face material bankruptcy risk. Without security, a future Chapter 11 filing could leave royalty holders with an unsecured claim.
Cap and term. Standard synthetic royalty financings include return caps (typically 1.5× to 3.0× invested capital) that limit the investor's total return. Since no capital was invested here, the concept of a return cap does not translate directly. The absence of a cap would mean the royalty continues indefinitely, or until patent expiry, a potentially extraordinary return for an instrument received in exchange for out-of-the-money warrants. The 5% rate is now the sole royalty burden on vidofludimus calcium following the 4SC settlement, meaning any acquirer or partner evaluating the asset faces a clean encumbrance profile — no stacking of legacy and synthetic royalties.
Change-of-control provisions. If Immunic is acquired before vidofludimus calcium reaches the market, what happens to the royalty? Standard synthetic royalty agreements include buyout provisions and change-of-control protections. An acquirer would need to either assume the royalty obligation or buy it out, creating a drag on acquisition economics similar to the HCR change-of-control payments I discussed in Royalties That Wake Up.
Accounting treatment. The cancellation of 51 million warrants will affect Immunic's balance sheet depending on whether those warrants were classified as equity or liabilities. If liability-classified (as many biotech warrants with fundamental transaction provisions are, per the ASC 815-40 analysis in The Warrant Sweetener), cancellation reduces total liabilities and potentially improves stockholders' equity, directly supporting the Nasdaq compliance objective. The issuance of royalty obligations creates a new off-balance-sheet commitment that will require disclosure but may not appear as a liability until the product generates revenue.
Transferability. Whether the royalty interests can be transferred to third parties will determine whether a secondary market can develop. If BVF Partners or other holders can sell their pro rata royalty shares, institutional royalty investors could aggregate small positions into meaningful economic interests. This would connect the warrant inducement market with the royalty financing market in ways neither currently anticipates.
Implications
For the Warrant Inducement Market
In The Warrant Sweetener, I documented the spectrum of warrant inducements from survival financing (Moleculin Biotech's serial inducements) through clinical-stage capital raises (SELLAS Life Sciences' $31 million transaction) to strategic restructurings (ADC Therapeutics / HCR). The Immunic transaction adds a new category to this spectrum: warrant retirement via royalty conversion. Unlike standard inducements, which require warrant holders to pay cash exercise prices and generate dilutive share issuances, the royalty conversion eliminates warrants entirely without any equity issuance or cash payment.
This matters most for companies whose stock prices have fallen so far below warrant exercise prices that standard inducements are impractical. As I showed in The Warrant Sweetener, Moleculin's serial inducements required progressively higher multipliers (2.0× to 3.0×) as its leverage deteriorated. Companies in Immunic's position, trading near $1.00 with warrants issued at $0.75, face a different constraint: the warrants are near enough to the money that they represent meaningful dilutive overhang, but the company's balance sheet cannot support paying cash consideration to retire them. A synthetic royalty on a late-stage asset fills this gap.
For Royalty Investors
The Immunic precedent creates a new class of synthetic royalty that did not exist before: micro-royalties originated through cap table restructurings rather than negotiated financings. These instruments may lack the protective covenants, security interests, and governance rights that characterise the institutional synthetic royalty deals tracked by Covington, Gibson Dunn, and Goodwin. But they also expand the investable universe. If transferable, these royalty interests could be aggregated by specialised funds, creating dealflow that originates in the equity markets rather than through bilateral negotiations.
For Acquirers
Any potential acquirer of Immunic must now account for a 5% royalty on vidofludimus calcium held by a dispersed group of former warrant holders, in addition to the standard equity, debt, and warrant obligations. The royalty will persist through any change of control unless specifically addressed in the acquisition agreement. This is the same structural complexity I flagged in The Warrant Sweetener regarding ADC Therapeutics, where HCR's warrants include deemed-exercise provisions in M&A scenarios. The difference is that the Immunic royalty is held by a potentially large number of counterparties, making a negotiated buyout more complex than a bilateral arrangement with a single institutional holder.
The Bigger Picture
The common thread connecting the ADC Therapeutics warrant issuance, the Karyopharm layered restructuring, the Immunic royalty swap, and the broader synthetic royalty market tracked by Covington and Goodwin is that pharmaceutical financing instruments are becoming fungible. A warrant that cannot be exercised for cash can be exchanged for a royalty that will not pay for years. A royalty agreement that blocks acquisitions can be amended in exchange for warrants that align the investor with equity outcomes. The instrument matters less than the economic exposure it delivers.
Royalty Pharma's $2 billion arrangement with Revolution Medicines in June 2025, combining a $1.25 billion synthetic royalty with a $750 million secured term loan, demonstrated that even the largest institutional transactions are becoming multi-instrument hybrids. The Immunic deal operates at the opposite end of the scale but illustrates the same principle: capital structure problems increasingly demand solutions that cross the traditional boundaries between equity, debt, and royalty instruments.
The question for the market is whether the Immunic structure remains an outlier or becomes a template. If vidofludimus calcium succeeds in Phase 3 and the royalty holders earn substantial returns, the playbook will be studied and replicated by every micro-cap biotech struggling with warrant overhang and a late-stage pipeline asset. If the drug fails, the exchange will be remembered as a clever footnote in a failed company's financing history. Either way, the structural innovation, using product economics to solve equity structure problems, will remain available to the next company that needs it.
All information in this report was accurate as of the research date and is derived from publicly available sources including company press releases, SEC filings, regulatory announcements, and financial news reporting. Information may have changed since publication. This content is for informational purposes only and does not constitute investment, legal, or financial advice.
Member discussion